Are The Bonds Between Polymer Molecules Weak Or Strong

Ever wondered what makes your favorite stretchy yoga pants so… well, stretchy? Or why that plastic bottle doesn't just crumble in your hand? It all boils down to something super cool happening at the tiniest level: the bonds between polymer molecules. Get ready to dive into the fascinating world of polymers, where "weak" and "strong" are more like playful dance partners than opposing forces.
Think of polymers like a gigantic necklace made of tiny, repeating beads. These beads are called monomers, and when they link up, they form a super long chain – that's your polymer! It's like a kilometer-long spaghetti noodle, but made of microscopic building blocks. This incredible length is what gives polymers their unique and often amazing properties.
The "Weak" Wonders: Gentle Hugs
Now, when we talk about the bonds between these long polymer chains, things get really interesting. Sometimes, the forces holding these chains together are pretty gentle. We call these intermolecular forces. Imagine them as little, friendly nudges or light hand-holds between the chains.
These "weak" bonds are like polite greetings at a party. They're there, they hold things together loosely, but they can be easily persuaded to let go. This is why some plastics, like the film wrap you use in the kitchen, can be so flexible and easily stretched. The chains can slide past each other a bit, thanks to those gentle nudges.
Think about taffy! You can pull it and stretch it, and it slowly, satisfyingly reshapes. That's the power of these more flexible, weaker bonds at play. They allow the material to deform without breaking. It's a kind of controlled flow, a delightful give-and-take.
Materials with these gentler bonds are often transparent or translucent. They let light pass through because the chains aren't packed in too tightly. It’s like looking through a lightly frosted window, where you can still see the shapes on the other side.
-min,h_400,q_80,w_640.jpg)
Even though we call them "weak," these forces are crucial. Without them, the polymer chains would just float around independently. They're the subtle glue that holds the structure together, allowing for that characteristic pliability that makes so many everyday items so useful and fun.
The "Strong" Superstars: Powerful Handshakes
But then, there are polymers with a much firmer grip! These have stronger bonds holding their chains together. Instead of light nudges, imagine these as firm handshakes or even a friendly, tight hug between the chains. These are also intermolecular forces, but they're the more robust kind.
When these forces are stronger, the polymer chains are held much closer together and can't slide past each other as easily. This makes the material much more rigid and resistant to deformation. Think about a hard plastic toy or the casing of your smartphone.
This is where we get into the realm of strength and durability. These stronger bonds mean the material can withstand more pressure and stress before it starts to bend or break. It's the difference between a flimsy plastic bag and a sturdy toolbox.

Consider a rock-hard LEGO brick. That solid feel comes from those powerful connections between the polymer chains. They are locked in place, giving the material its impressive toughness. It's a testament to how molecular structure dictates macroscopic properties.
These strong bonds can also affect how materials behave with heat. Polymers with strong intermolecular forces often have higher melting points. They need a lot more energy to get those chains moving freely enough to become liquid.
The Magic Mix: It's Complicated (In a Fun Way!)
Here's where it gets really entertaining: most polymers aren't just "weak" or "strong." They're often a fascinating mix of both! It's like having a group of friends where some give gentle pats and others enthusiastic bear hugs.
The specific type of monomer and how the polymer chains are arranged make all the difference. Sometimes, you have chains that are super long and tangled, like a massive ball of yarn. Even with weaker bonds, this entanglement can provide significant strength. The sheer amount of "stuff" involved creates resistance.

Then there are polymers where some sections of the chains are drawn to each other more strongly than others. Imagine a chain with little magnets dotted along its length. These magnetic bits will attract other magnets on neighboring chains, creating strong spots.
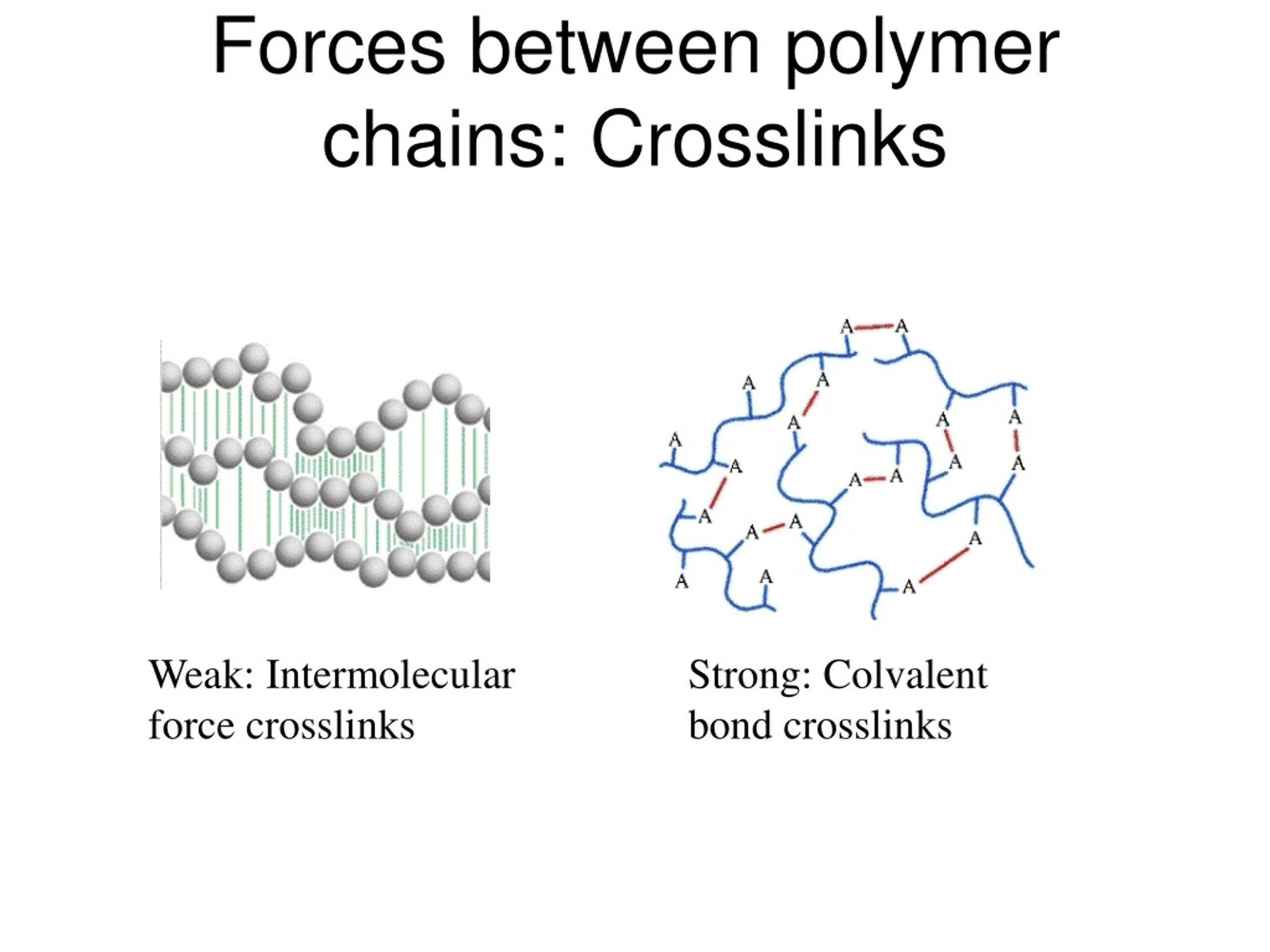
And what about cross-linking? This is a super exciting concept! Here, some chains are actually chemically bonded to other chains, creating a three-dimensional network. Think of it like weaving a fabric instead of just laying threads side-by-side.
These cross-links are like super-strong, permanent welds between the polymer chains. They make the material incredibly tough, elastic, and resistant to dissolving. This is how we get things like rubber and many advanced plastics.
The famous vulcanization of rubber, a process discovered by Charles Goodyear, is a prime example. It introduces sulfur cross-links into rubber, transforming it from a sticky, meltable mess into the durable, bouncy material we know and love. It’s a classic science story of transformation!
Why Should You Care (Besides Your Yoga Pants)?
Understanding these bonds is like having a secret key to the material world. It explains why we have everything from soft, fluffy blankets to hard, durable helmets. It's the science behind innovation!
Scientists and engineers play with these molecular bonds like a puzzle. They can design polymers for specific jobs. They can make materials that are lightweight yet incredibly strong, or flexible yet waterproof. The possibilities are truly endless!
This knowledge helps us create better products. Think about lighter airplanes that save fuel, stronger medical implants that improve lives, or more sustainable packaging that reduces waste. It’s all thanks to mastering these molecular interactions.
So, the next time you stretch your favorite t-shirt or marvel at the strength of a plastic container, remember the incredible dance of the polymer chains. Whether it's a gentle hug or a firm handshake, these molecular bonds are the unsung heroes of our everyday lives, making the world around us functional, flexible, and frankly, pretty amazing. It’s a little bit of molecular magic, and it’s all around you!
