Balanced Equation For Sodium Hydroxide And Sulfuric Acid
Alright, pull up a chair, grab your latte, and let’s chat about something that might sound as exciting as watching paint dry, but trust me, it’s got more fizz and bang than you’d expect. We're diving into the thrilling world of… wait for it… chemical reactions! Specifically, the spectacular showdown between sodium hydroxide and sulfuric acid. Think of it as a dramatic love story, or maybe a really intense wrestling match, between two molecular heavyweights.
Now, I know what you’re thinking. "Chemical reactions? Isn't that for nerds in lab coats with way too much hair gel?" And to that, I say, bless your heart. But even the simplest chemical reactions are like tiny, invisible fireworks happening all around us, and this one? This one’s a real showstopper. It’s the kind of reaction that makes you say, "Whoa, molecules can do that?"
First up, let’s meet our contestants. On one side, we have sodium hydroxide. You might know it by its more common, and frankly, much cooler, name: caustic soda. Don’t let the fancy name fool you; this stuff is basically the industrial-strength cleaning lady of the chemical world. It’s a super-strong base, which means it loves to grab onto pesky little acid molecules and neutralize them. It’s like the ultimate chill pill for acids. Imagine a really, really grumpy dude who’s been arguing with everyone all day, and then along comes this super-calm person who just says, "Hey, relax, have a cookie," and suddenly, he's all smiles. That’s sodium hydroxide for a sulfuric acid.
On the other side, we’ve got sulfuric acid. This one’s a bit of a diva. It’s a super-strong acid, and it can be, shall we say, a little enthusiastic in its reactions. Think of it as the party animal of the periodic table. It’s got a reputation for being a bit aggressive, and honestly, it’s earned it. It’s used in everything from making fertilizers (which, by the way, are basically how we feed the world without everyone resorting to gnawing on tree bark) to refining oil. This stuff is powerful, and sometimes, a little bit scary if you’re not careful. It’s the kind of chemical that makes you want to wear safety goggles, a lab coat, and possibly a hazmat suit, just in case. But in the right hands, it’s a miracle worker!
So, what happens when these two titans collide? Well, it’s a beautiful thing, really. It’s a classic acid-base neutralization reaction. It’s like a cosmic dance where they find common ground and, in the process, create something new and, dare I say, useful. They stop being their grumpy, diva selves and become something… well, something much more serene.

The magic happens when you mix them together in just the right amounts. This is where our fancy balanced chemical equation comes in. Now, don't let the word "equation" intimidate you. It's just a way scientists write down what's happening in a chemical reaction, like a recipe for molecular madness. It tells us exactly how many of each ingredient (molecule) we need to get the perfect result. Think of it as the cheat sheet for making the universe work.
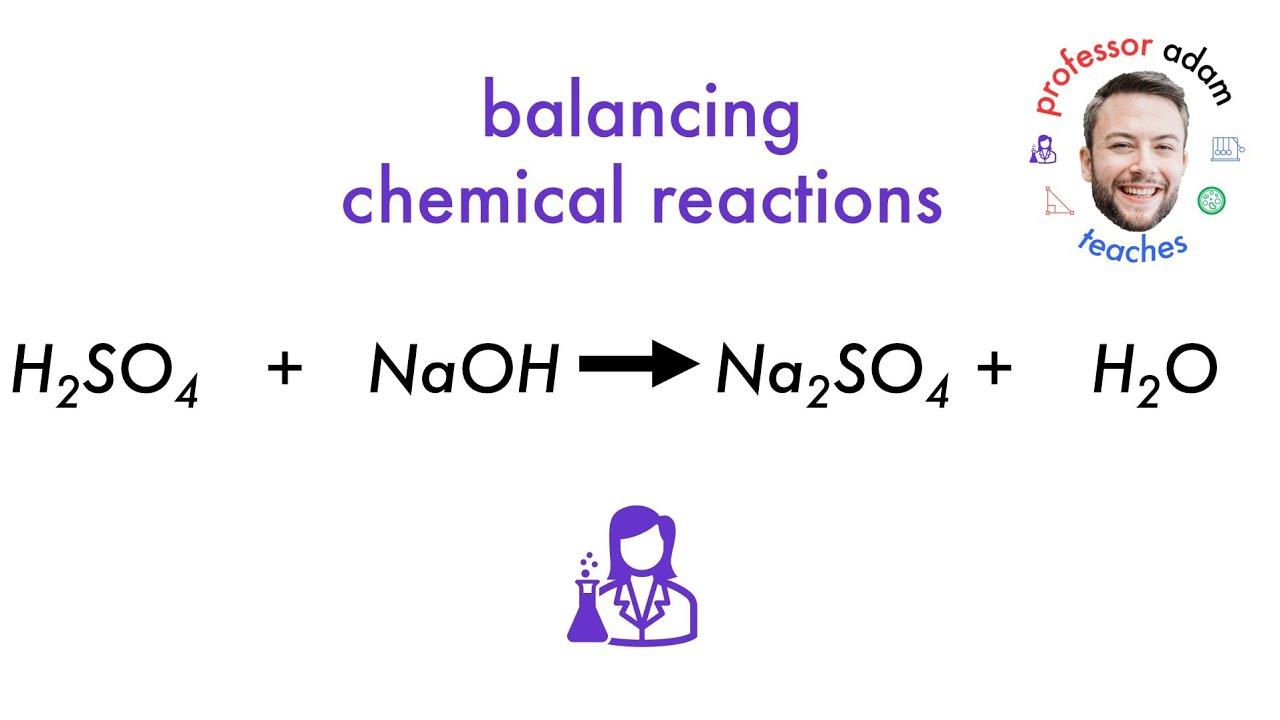
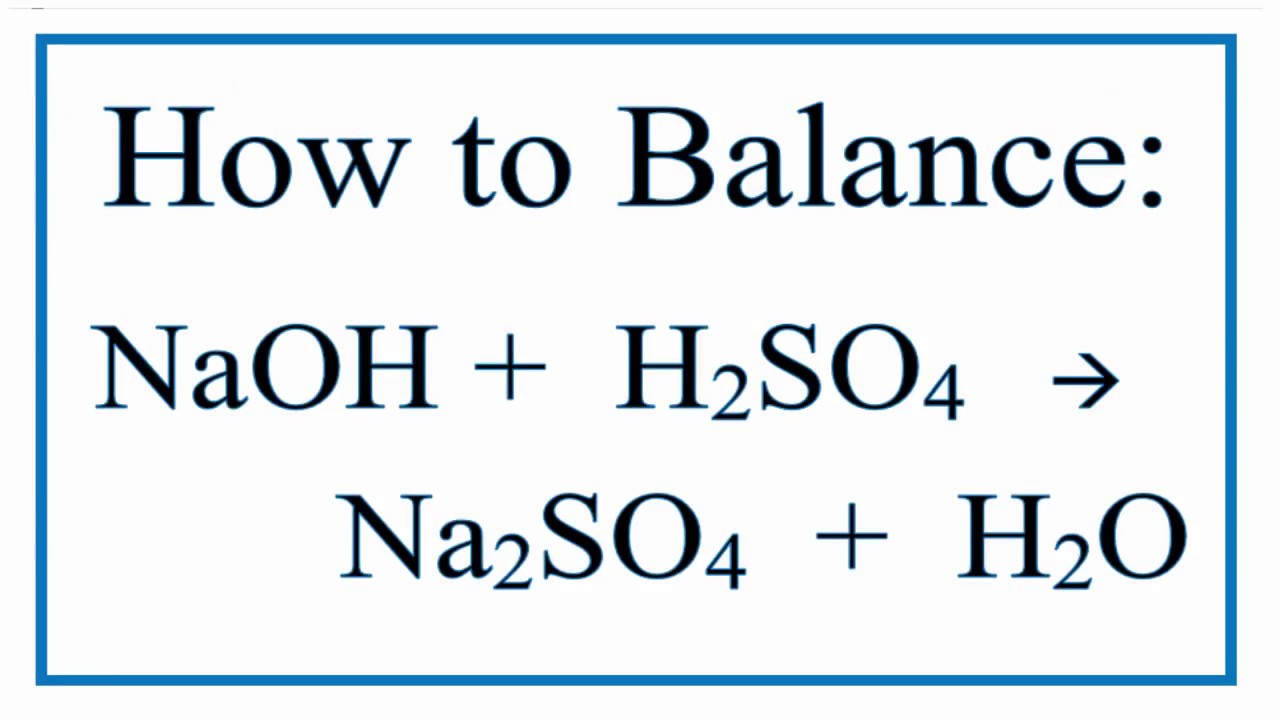
Here’s the equation, in all its glory:
2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
Let’s break this down, shall we? It’s not as intimidating as it looks. Seriously, it’s like deciphering a secret code, but the reward is knowing how the universe actually works. Pretty neat, right?
On the left side, we have our reactants – the guys who are about to get together. We've got 2NaOH. See that big '2'? That means we need two molecules of our friendly, neighborhood sodium hydroxide. It’s like needing two scoops of ice cream to make a really awesome milkshake.
Then we have H₂SO₄. That’s our sulfuric acid, the party animal. This time, we only need one molecule of it. It’s like needing just one shot of espresso to kickstart that milkshake into high gear. So, it’s two sodium hydroxides to one sulfuric acid. It's all about balance, people!

Now, for the arrow (→). In the chemical world, this is like saying, "And then… BAM!" or "And the result is…". It's the moment of transformation, the cliffhanger in our molecular soap opera.
And on the right side? That’s where the magic (or the science) happens. We get Na₂SO₄. This is sodium sulfate. Think of it as the chill, happy byproduct of this whole ordeal. It’s completely neutral, no longer angry or diva-like. It's the perfectly blended smoothie that comes after all the ingredients have done their thing. It's surprisingly useful, too – used in detergents and glass manufacturing. Who knew a bit of molecular drama could lead to cleaner dishes?
And the other product? That’s 2H₂O. You know what that is, right? That’s right, water! Two molecules of it, to be exact. So, this intense acid-base reaction, this molecular wrestling match, ultimately produces… water and a salt. Isn't that just the most anticlimactic yet beautiful thing you've ever heard? It's like all that energy and drama just ends with a refreshing glass of water and a sprinkle of salt. It’s the universe saying, "See? We can make something peaceful out of chaos." It’s a powerful reminder that even the most potent forces can be tamed and transformed into something gentle and life-sustaining. Mother Nature, you’re a poet!

The reason this equation is balanced is that the number of atoms of each element is the same on both sides. If you count up all the sodium atoms on the left, you’ll find the same number on the right. Same for hydrogen, sulfur, and oxygen. It's like making sure you have the same number of cookies in the jar before and after you share them. No cheating allowed in the chemical kitchen!
This whole process is super important in so many industries. It’s how we control the acidity of things, how we create different compounds, and how we make sure that our cleaning products are effective without, you know, dissolving your entire house. It’s the unsung hero behind many of the things we take for granted.
So next time you hear about sodium hydroxide and sulfuric acid, don't just think of scary chemicals. Think of the balanced equation, the molecular dance, the creation of sodium sulfate and, yes, good old water. It’s a little bit of everyday magic, happening right under our noses. Now, who wants another coffee? I feel like a chemistry professor after all that.
