Balanced Equation Of Sodium Hydroxide And Sulfuric Acid
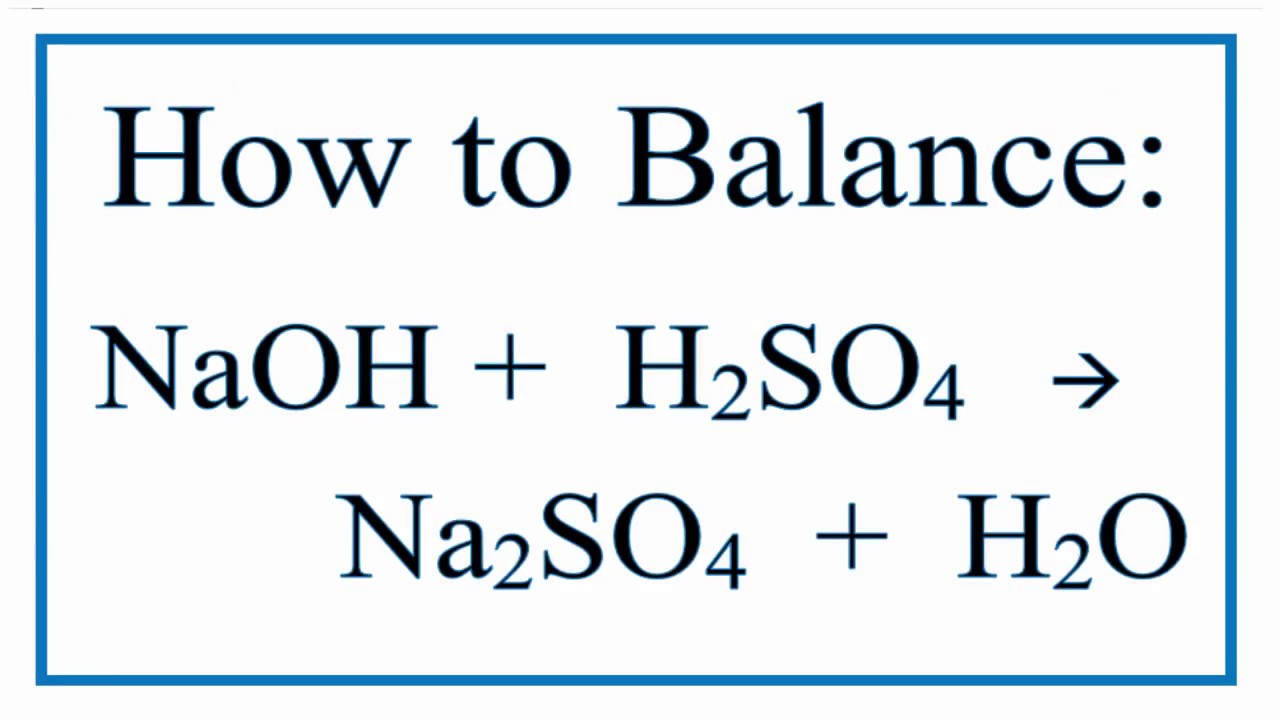
You might not think a chemical reaction could be a source of creative inspiration, but the balanced equation of sodium hydroxide and sulfuric acid is surprisingly popular in certain circles, often showcasing a delightful dance of neutralization that can be quite visually appealing! It’s not about explosions or dramatic transformations, but rather a quiet, elegant process that’s captured the imaginations of many. Think of it as the art of controlled chemistry, where precision meets predictable beauty.
For artists, hobbyists, and anyone with a curious mind, dabbling in this seemingly simple equation can unlock a world of possibilities. Imagine a painter using the subtle color shifts that can occur with slight variations in concentration as inspiration for a pastel palette. Or a sculptor finding a new appreciation for form and structure by observing the precise way reactants combine. For casual learners, it’s a fantastic way to demystify a fundamental chemical concept. Understanding how an acid and a base react to form salt and water isn't just educational; it's about building confidence in exploring the world around you. It’s about realizing that science can be accessible and even beautiful.
The variations are as diverse as the individuals trying it. Some experiment with different concentrations to witness subtle changes in temperature or viscosity. Others might focus on the visual aesthetics of the precipitation, if any occurs, creating patterns or textures. Think of the clean lines of a perfectly neutralized solution, or the delicate swirl of reactants coming together. You could even conceptualize this reaction in abstract art, representing the balancing act of opposing forces. The subject matter is the reaction itself, but the style can range from hyperrealistic depictions of laboratory equipment to more metaphorical interpretations of harmony and equilibrium.
If you’re eager to try this at home, remember that safety is paramount. Always wear appropriate protective gear like gloves and eye protection, and ensure good ventilation. Start with small, diluted quantities. You can find basic kits online or at educational supply stores that will provide you with the necessary materials and clear instructions. It's a wonderful way to engage with chemistry in a tangible, hands-on manner, without the need for advanced laboratory setups. Think of it as a culinary experiment for the scientifically inclined, where precise measurements lead to a fascinating outcome.
Ultimately, the joy of exploring the balanced equation of sodium hydroxide and sulfuric acid lies in its ability to blend intellectual curiosity with a touch of artistry. It’s a testament to the elegance of chemical principles and the infinite ways we can observe and appreciate them. It's about the satisfaction of understanding a fundamental process, and perhaps, finding a new appreciation for the precise and often understated beauty that exists within the world of science. It's an invitation to look closer, to measure carefully, and to discover the quiet wonder of a world in balance.
