Balanced Equation Of Sulphuric Acid And Sodium Hydroxide

Hey there, ever felt like you’re trying to perfectly balance two things in your life? Maybe it's work and play, or that last slice of pizza and your diet? Well, guess what? The world of chemistry has its own balancing acts, and one of the most important ones involves something you might have heard of: sulfuric acid and sodium hydroxide. Don’t let the fancy names scare you off! Think of them like two strong personalities that, when they meet under the right conditions, create something wonderfully calm and useful.
Let's break it down in a way that’s as easy as making your morning coffee. We’ve got sulfuric acid, which is a bit of a fiery, energetic character. Imagine it like a super-powered cleaning agent, the kind that can really get into the tough grime. It’s a strong acid, meaning it’s got a real oomph to it. On the other side, we have sodium hydroxide. This one’s a strong base. Think of it like a super-soother, something that can calm down a lot of energetic reactions. It’s also known as lye, and it’s been used for centuries to make soap – so, pretty important stuff!
Now, when these two get together, they don’t just go wild. They have a very specific way of interacting, and this is where our balanced equation comes in. It’s like a recipe, showing exactly how much of each ingredient you need to get the perfect result. No more, no less. It’s all about harmony and making sure everything is just right.
The Big Meet-Up: Acid Meets Base!
Imagine you’re having a bit of a chaotic party. You’ve got the loud music, the kids running around, everyone’s super excited and maybe a little too energetic. That’s kind of like our sulfuric acid – it’s got a lot of energy, a lot of protons to give away. Now, imagine you introduce a bunch of calm, collected adults who are great at listening and soothing things down. That’s our sodium hydroxide, ready to absorb all that extra energy.
When sulfuric acid (H₂SO₄) and sodium hydroxide (NaOH) meet, they perform a chemical dance. The acid has a surplus of hydrogen ions (H⁺), which are like little energetic sparks. The base, sodium hydroxide, has hydroxide ions (OH⁻), which are like little sponges, just waiting to grab those sparks. It’s a classic acid-base neutralization reaction. The goal? To get rid of that intense energy and create something much more… well, neutral.
Think about it like this: You’ve got a really sour lemon (acid) and a spoonful of baking soda (a base, sort of like sodium hydroxide in this context). When you mix them, you get that fizzing, bubbling action, right? That fizzing is the energetic reaction settling down, becoming less intense. Sulfuric acid and sodium hydroxide do something similar, but in a much more controlled and powerful way.
The Recipe: What Does the Equation Tell Us?
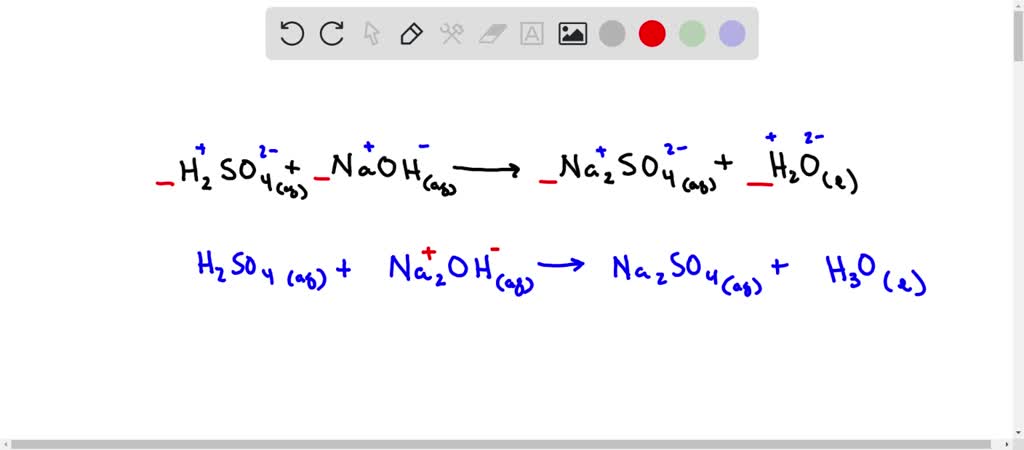
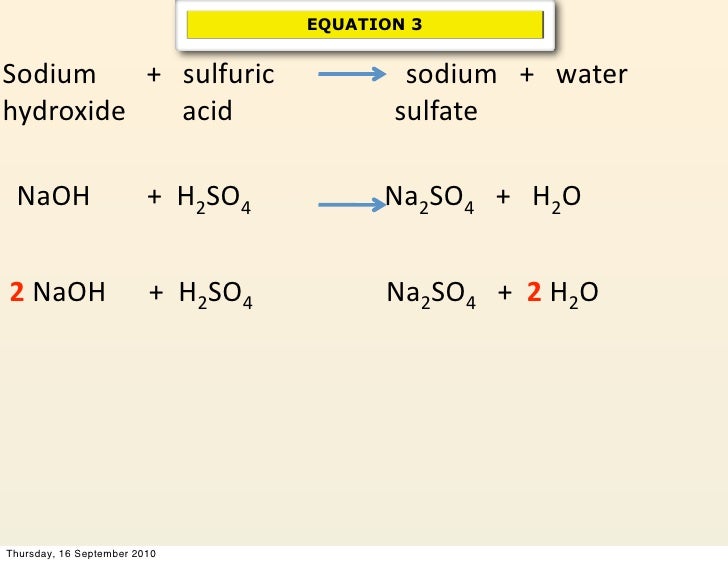
Here’s the magic formula, the balanced equation:
2NaOH + H₂SO₄ → Na₂SO₄ + 2H₂O
Now, let’s translate that from chemistry speak to everyday language. It’s like saying:
“For every one molecule of sulfuric acid, you need two molecules of sodium hydroxide to make things perfectly peaceful.”

The stuff on the left side (2NaOH + H₂SO₄) is what you start with – your reactants. These are the energetic personalities before they’ve had a chance to chat and calm down. The stuff on the right side (Na₂SO₄ + 2H₂O) is what you end up with – your products. These are the calm, happy results of their interaction.
Na₂SO₄ is sodium sulfate. Think of this as the happily married couple, the byproduct of their union. It’s a salt, and these guys are pretty stable and often quite useful. In this case, it’s a pretty common salt.
And then there’s H₂O. Yep, you guessed it! That's water. The universal solvent, the ultimate peacekeeper. The reaction of a strong acid and a strong base almost always produces water. It’s like the final sigh of relief after a big storm, everything is calm and clean.
Why Should We Even Care?
Okay, so we’ve got chemicals doing their thing. Why should you, sitting there perhaps with a cup of tea or scrolling through your phone, care about this particular chemical dance?
Well, this seemingly simple reaction is the backbone of a lot of things that make our modern lives possible. Think about it:
Cleaning Power: Sulfuric acid, in carefully controlled amounts and dilutions, is used in many cleaning products, especially for heavy-duty tasks like unblocking drains. But it's way too strong to use by itself! Sodium hydroxide is also a powerful cleaner and a key ingredient in making soap and detergents. Understanding how they neutralize each other helps us handle these substances safely and effectively, so they can do their cleaning jobs without causing harm.
Water Treatment: Believe it or not, this neutralization concept plays a role in purifying our water. Sometimes, industrial wastewater can be too acidic. Adding a base, like sodium hydroxide, can help bring the pH back to a safe level before it’s released back into the environment. It’s like giving a grumpy, over-caffeinated river a nice, calming drink.
Making Stuff: From fertilizers that help grow our food to the processing of paper and textiles, sulfuric acid is a massive industrial chemical. Sodium hydroxide is just as vital, used in everything from making aluminum to producing rayon. These reactions, precisely balanced, are crucial for producing the materials we rely on every single day. Imagine trying to make your favorite cotton t-shirt without balanced chemistry – it just wouldn’t happen!

Safety First: Knowing that these strong chemicals neutralize each other is also about safety. If you accidentally get a bit of sulfuric acid on your skin, applying a weak base (like baking soda solution, though in industrial settings they'd use specific procedures) can help neutralize it and reduce the damage. This is a basic principle of first aid for chemical spills. It’s like knowing that a gentle pat on the back can calm down an overexcited friend – in this case, a much more serious situation!
The Little Story of Balance
Let me tell you a little story. Imagine a tiny, bustling chemical factory. Inside, there are two main machines. One machine, the “Acid Dispenser,” churns out lots and lots of energetic H⁺ ions. It’s so busy, it’s practically vibrating! Next to it is the “Base Balancer,” which produces OH⁻ ions, calmly waiting to do its job.
If the Acid Dispenser just kept pumping out ions without anyone to balance them, the whole factory would become a chaotic mess, too harsh for anything to function. But when the Base Balancer steps in, it carefully takes two OH⁻ ions for every H₂SO₄ molecule that comes its way (remember the 2:1 ratio!). They link up, forming stable Na₂SO₄ molecules and little droplets of H₂O. The factory floor becomes clean, orderly, and ready for more work. It’s a perfect example of how balance, even in the smallest of things, leads to productivity and harmony.
So, the next time you hear about sulfuric acid and sodium hydroxide, don’t just think of them as intimidating chemical names. Think of them as two powerful forces that, when balanced correctly, create the very building blocks of our world and keep things running smoothly, safely, and cleanly. It’s a chemical hug, a neutralization hug, that makes life just a little bit better for all of us!
