Clay Like Substance From Which Aluminium Is Obtained

Imagine your favorite shiny, lightweight soda can. Or maybe that super-strong yet feather-light bicycle frame that lets you zoom up hills like a superhero. Ever wondered where all that amazing aluminum comes from? Well, prepare for a bit of a surprise, because it doesn't start in a gleaming metal mine. Nope, our friend aluminum begins its journey buried deep in the earth, hidden inside something that looks suspiciously like… well, dirt!
More specifically, it’s found in a rock called bauxite. Now, "bauxite" might sound like some exotic, far-off gem, but think of it more like the earth’s slightly grumpy, reddish-brown clay. It’s not exactly pretty, and you certainly wouldn’t want to build a sandcastle out of it. But within this unassuming, dusty stuff lies a secret treasure trove of the metal that makes our lives so much easier and cooler.
Think about it: we use aluminum for so many things! It’s in airplanes, letting us soar through the skies. It’s in our kitchens, protecting our food and making cooking a breeze with those trusty baking pans. Even your smartphone, that little rectangle of magic, has aluminum keeping its delicate innards safe and cool. And it all starts with this humble, earthy material.
The story of how we get from dusty red dirt to sparkling metal is quite a tale. It involves some clever science and a whole lot of effort, but at its heart, it's a story of transformation. Imagine this: scientists and engineers looking at this ordinary-looking rock and saying, "You know what? There's something special in there!" It’s like discovering a hidden talent in your quietest friend.
The process of extracting aluminum from bauxite is called the Bayer process. Don't let the fancy name scare you! Think of it like a very complicated recipe. First, they grind up the bauxite until it's a fine powder. Then, they mix it with hot, caustic soda. Now, caustic soda sounds a bit scary, but in this context, it acts like a super-powered solvent. It’s like using a powerful degreaser to get rid of stubborn grime, but on a much bigger scale.
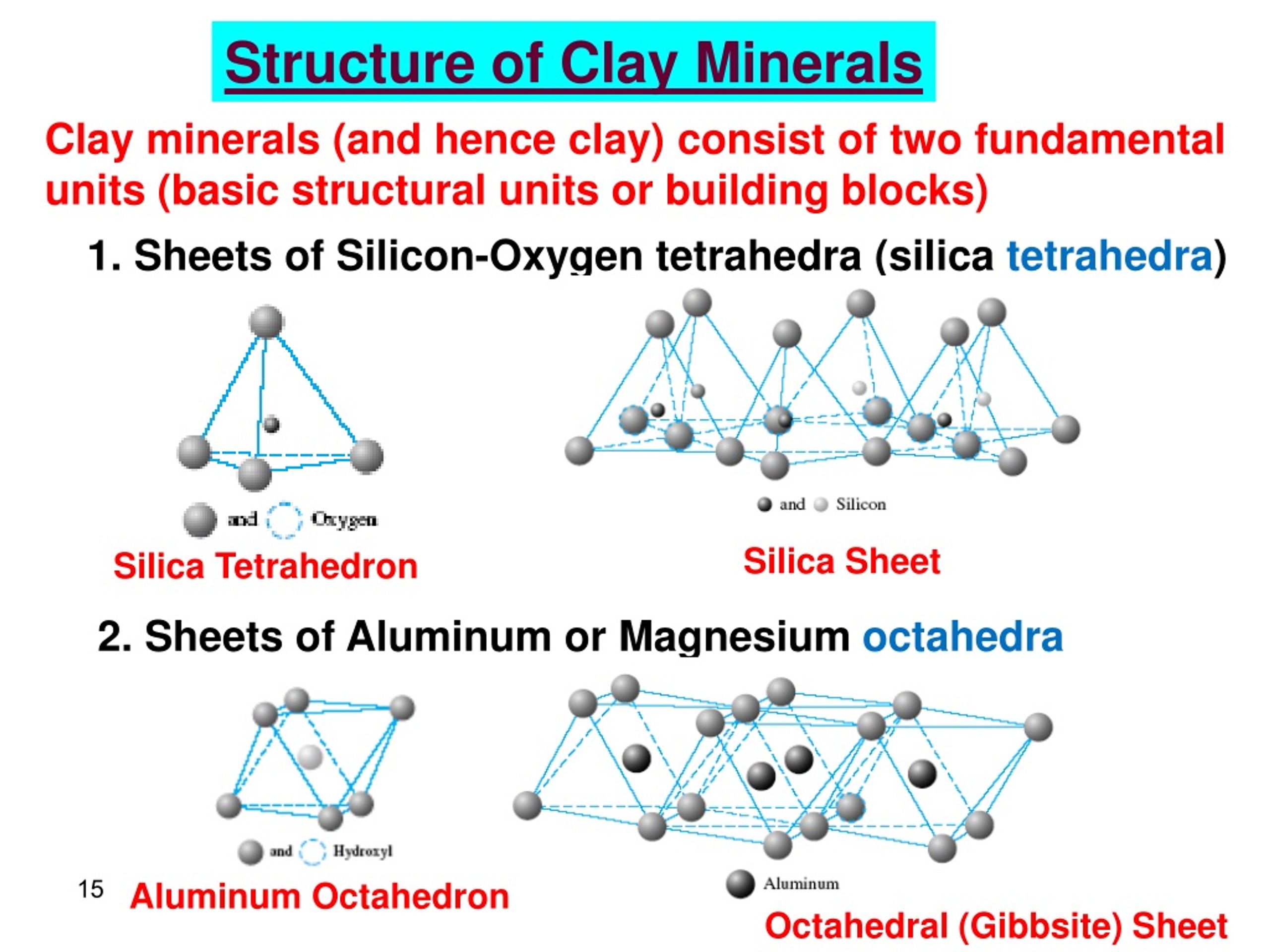
This mixture is then heated and stirred, and believe it or not, the aluminum actually dissolves, leaving behind all the other unwanted bits of the rock. It's like magic, but it's science! The liquid that’s left is a bit like a super-concentrated aluminum soup. This "soup" is then cooled, and guess what? Pure aluminum oxide crystals start to form. These crystals are then filtered out, looking a bit like white sand.
But we’re not quite at shiny metal yet. This white powder, aluminum oxide (also known as alumina), still needs a bit more coaxing. This is where things get really interesting and, dare I say, a little bit dramatic. Alumina is incredibly tough to break apart. It’s like trying to pull apart two super-glued best friends who refuse to be separated. To do this, we use a process called electrolysis.

Electrolysis is where electricity comes into play. Imagine a giant vat, kind of like a swimming pool, filled with molten alumina and another chemical called cryolite. Cryolite is important because it lowers the melting point of alumina, making it easier to work with. Then, they plunge these massive carbon rods, called electrodes, into the molten mix. When electricity flows through these electrodes, it’s like a powerful jolt that forces the aluminum atoms to let go of their oxygen buddies.
It's like a high-voltage breakup, but for atoms!
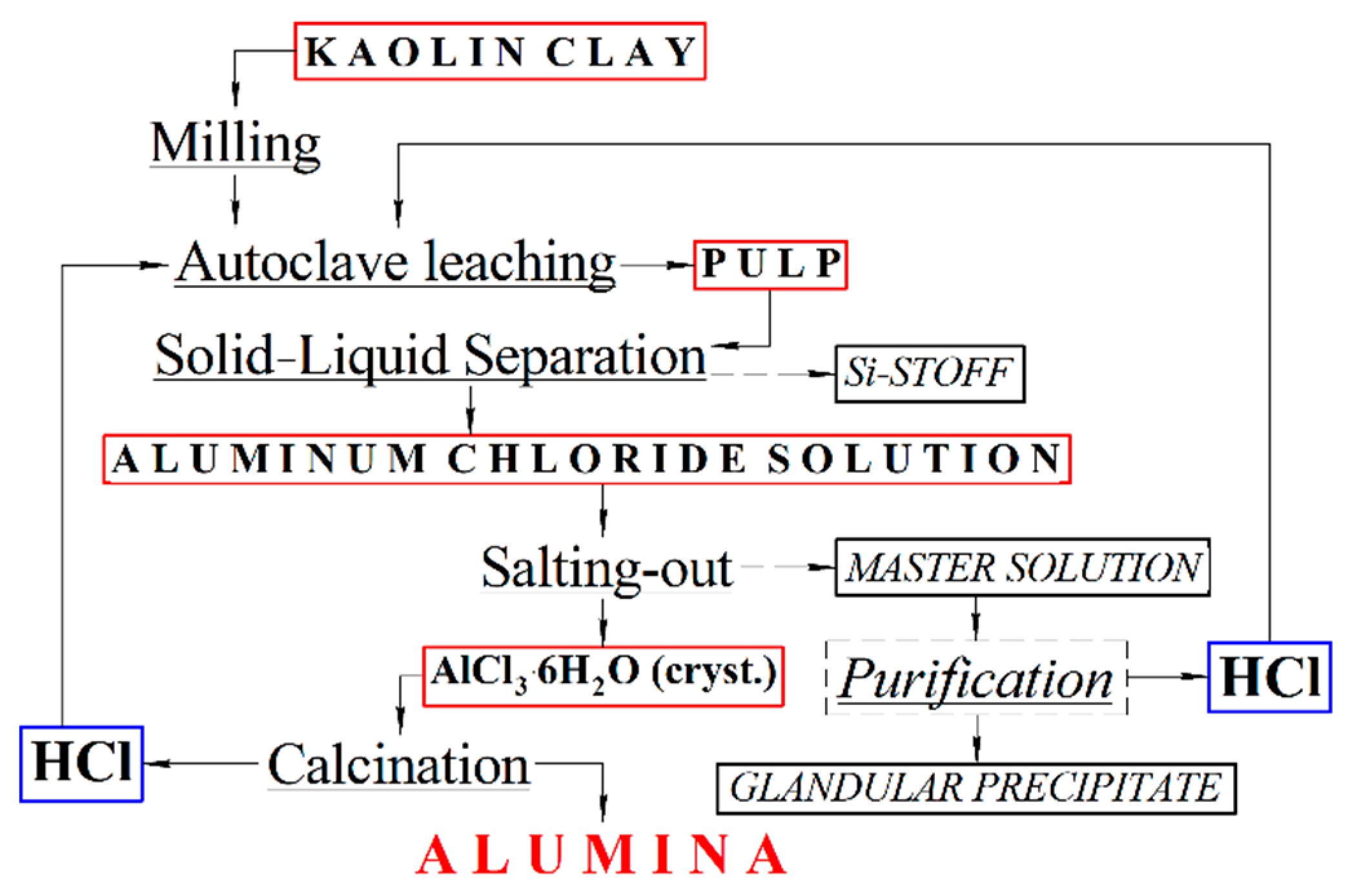
Obtaining Alumina from Kaolin Clay via Aluminum Chloride
The pure, molten aluminum, being heavier, sinks to the bottom of the vat, while the oxygen atoms are grabbed by the carbon electrodes and turn into carbon dioxide gas. And there it is! Molten aluminum, ready to be tapped off and poured into molds. It’s a dazzling, fiery spectacle, a testament to human ingenuity turning a humble, earthy material into something so incredibly useful and ubiquitous.
So, the next time you pop open that soda can or admire your sleek laptop, take a moment to appreciate the journey of that aluminum. It's come a long way from its humble beginnings as dusty, reddish clay. It’s a reminder that sometimes, the most amazing things are hidden in plain sight, just waiting for us to discover their incredible potential. And that, my friends, is a pretty heartwarming thought, don't you think?