Describe How Crude Oil Is Separated Into Fractions

Imagine you’ve just unearthed a treasure chest overflowing with a goopy, black, magical goo! That, my friends, is pretty much what crude oil is like. It’s this amazing, murky liquid that the Earth has been hiding away, and when we find it, it’s like… well, it’s like finding a secret ingredient for pretty much everything cool in our modern world. But here’s the kicker: this goopy treasure isn’t just one thing. Nope, it’s actually a whole bunch of different things all mixed up, like a super-sized, prehistoric smoothie. And our job, the super fun job, is to sort out this smoothie into its yummy, useful parts.
Think of it like this: you have a giant bowl of trail mix. You’ve got peanuts, raisins, chocolate chips, and maybe some weird little dried fruit things you can’t quite identify. Crude oil is kinda the same, but instead of yummy snacks, it’s made up of different-sized molecules. Some are tiny little things, like zippy little sprites, and some are huge, lumbering giants. And guess what? These different-sized molecules have different personalities, which means they do different jobs!
So, how do we get these tiny sprites and giant molecules to stop hogging the same party space? We send them on a rollercoaster ride, but not just any rollercoaster. This one is called fractional distillation, and it’s basically a fancy way of saying we’re going to use heat and a super-tall tower to sort them out. Imagine our crude oil smoothie is poured into a giant, fiery cauldron – don’t worry, it’s all very controlled and science-y! This cauldron heats the oil up, and when things get hot, those zippy little molecules get super excited and turn into a gas. It’s like they’re so happy to escape the sticky goo that they just… poof! Float away.
Now, this gas, which is now full of our teeny-tiny, energetic molecules, floats up into this enormous tower. This tower is the star of the show, and it’s called a fractionating column. Think of it as a skyscraper designed specifically for sorting molecules. It’s got lots of floors, or trays, at different heights. And here’s the cool part: the higher up you go in the tower, the cooler it gets. It’s like a natural air conditioner for our gas! Our super-excited gas starts to cool down as it rises.
And just like how water vapor turns back into water when it cools, our different-sized molecules start to chill out and condense back into liquids at different temperatures. The smallest, lightest molecules, the ones that are super energetic even when they’re cool, they stay as a gas all the way at the very top of the tower where it’s the chilliest. These are the superstars that become things like petroleum gas, which you might know as the stuff that powers your barbecue or your heater. They’re the zippiest, zingiest parts of the crude oil!
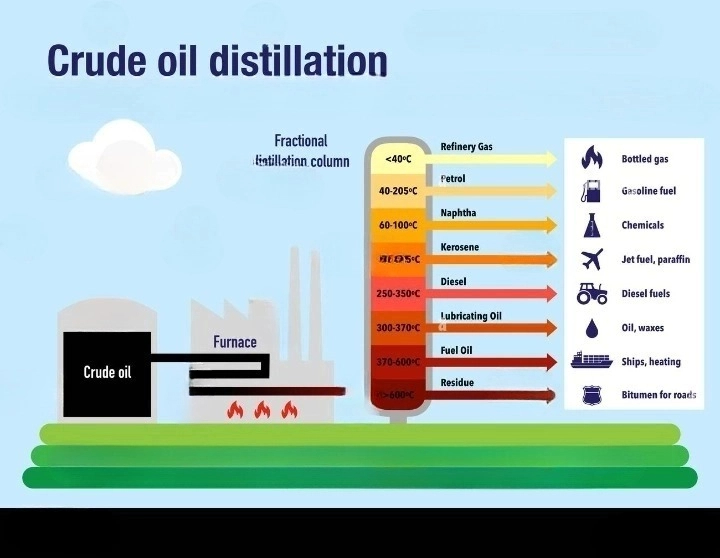
As you go down the tower, it gets warmer. So, the next size of molecules, a bit chunkier than the gas sprites, they start to cool down and condense on the trays a little further down. These guys are pretty important too! They become things like gasoline (or petrol, if you’re on the other side of the pond) – the super fuel that makes our cars zoom! Imagine all those road trips and adventures, all powered by these slightly bigger, but still pretty speedy, molecules.
Keep going down, and you’ll find even bigger molecules. These are the ones that are a bit more chilled out, they need a bit more warmth to stay in their liquid form. They become things like kerosene, which is used in jet engines (yes, the big metal birds that fly us to exotic places!) and also in things like lamp oil. Then you have diesel fuel, which powers those big trucks and buses that keep our cities running. These molecules are a bit more robust, ready for the heavy lifting.
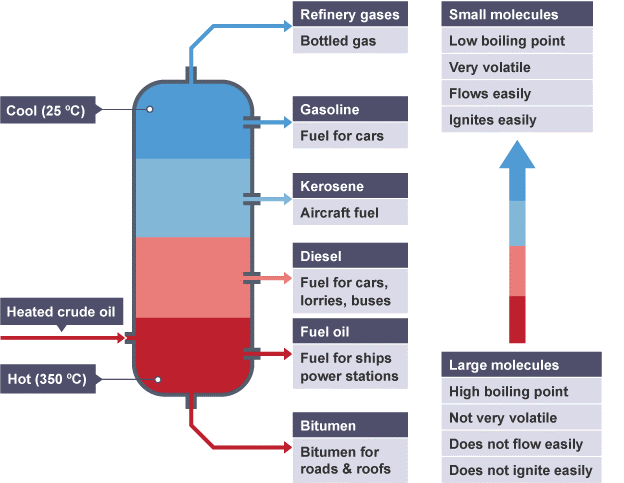
And then there are the really big, heavy molecules. These guys don’t even want to be gases at all! They’re like the comfy, couch-potato molecules of the crude oil world. They condense at the bottom of the tower, where it’s the hottest. These are the ones that become things like lubricating oil, which keeps your bike chain smooth and your car engine from grumbling sadly. And at the very, very bottom, after everything else has been collected, you’re left with the thickest, gooiest stuff, like bitumen. This is what we use to build our roads – those smooth surfaces we drive on every day! It’s like the foundation of our transportation system.
So, this whole process, this magical sorting of the crude oil smoothie, is called fractional distillation. We take this one, murky, mysterious liquid and, with a bit of heat and a super-tall tower, we break it down into all sorts of useful fractions, or parts. It’s like unlocking a treasure chest and finding not just one gem, but a whole collection of different, incredibly valuable gems, each with its own special sparkle and purpose. It’s a fantastic feat of science that gives us so much of what we use and rely on every single day!
