Describe The Arrangement Of Particles In A Solid

Ever looked at a sturdy brick wall and wondered what’s really going on in there? Or maybe you've admired the intricate design of a snowflake, or the dependable heft of your favorite coffee mug. These aren't just random chunks of stuff; they're marvels of molecular architecture! Today, we're diving into the incredibly organized, surprisingly neat world of solids and how their tiny building blocks, the particles, are arranged. Think of it as the ultimate minimalist interior design, but on a microscopic scale.
We often take solids for granted. They’re the bedrock of our existence, from the chair you're sitting on to the phone in your hand. But beneath that solid surface lies a universe of order. Unlike the wild and free-wheeling particles in gases or the gracefully sliding ones in liquids, solids have a serious commitment to structure. They’re not just hanging out; they’re locked in, maintaining a rather respectable distance from each other and holding onto their shape with admirable tenacity.
The Party in the Crystal Palace: Crystalline Solids
Let's start with the rockstars of the solid world: crystalline solids. These are your diamonds, your salt crystals, your snowflakes, even the sugar in your tea. In these materials, particles aren't just jumbled together; they’re arranged in a highly predictable, repeating pattern. Imagine a meticulously planned dance floor where every dancer knows their exact position and cue. This repeating, three-dimensional pattern is called a crystal lattice.
Think of it like a perfectly laid out mosaic tile floor, but in all three dimensions. Each tiny particle – whether it’s an atom, an ion, or a molecule – is nestled into its designated spot. They’re not moving around freely; instead, they’re essentially vibrating in place. It’s like being at a very polite, very organized party where everyone’s doing a little jig in their spot but not actually leaving it. This vibration is key; it’s what gives solids their characteristic rigidity.
There are different types of crystal lattices, depending on what kind of particles are involved and how they bond. For instance, ionic crystals, like table salt (NaCl), have positively charged sodium ions and negatively charged chloride ions arranged in an alternating pattern. They're held together by strong electrostatic attractions, like tiny magnets pulling on each other. This gives them a strong, brittle structure – ever dropped a salt crystal? It shatters, but in a way that still reveals its organized nature.
In metallic crystals, like a copper wire or a gold ring, metal atoms are packed closely together. The electrons in their outer shells are not tied to any single atom; instead, they form a “sea” of electrons that flows around the metal ions. This is what makes metals such excellent conductors of electricity and heat – those little electrons are free to move and carry the energy around! It’s like having a super-efficient delivery service within the solid structure.
Molecular crystals, like ice (solid water) or sugar, are made of discrete molecules. These molecules are held together by weaker forces, like Van der Waals forces or hydrogen bonds. While the molecules themselves are intact, the forces between them are not as strong as ionic or metallic bonds. This is why ice can melt into liquid water without its molecules breaking apart – the weak intermolecular forces are overcome, but the strong covalent bonds within each H₂O molecule remain intact. It's like a group of friends holding hands; they can let go of each other's hands and mingle, but they're still themselves.
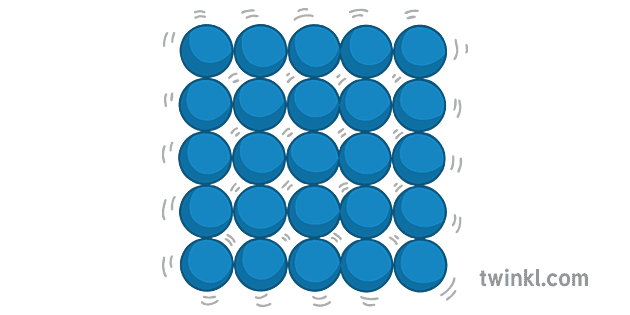
Fun Fact: The iconic structure of a snowflake isn't just beautiful; it’s a testament to the precise arrangement of water molecules in ice. Each arm of the snowflake is a hexagonal crystal, reflecting the underlying hexagonal lattice structure of ice. The unique patterns arise from tiny variations in temperature and humidity as the snowflake forms in the atmosphere.
Practical Tip: Admire Your Crystals!
Next time you have a salt shaker or a sugar bowl, take a moment to look closely. Can you see the tiny, often cubic, crystal structures? Even the quartz crystal in your watch is a perfectly ordered arrangement of silicon and oxygen atoms. It’s a little piece of microscopic art in your everyday life.
The Slightly Messy Studio Apartment: Amorphous Solids
Now, not all solids are as rigidly organized as a crystalline palace. Some are more like a well-loved, slightly chaotic studio apartment – still functional, still a solid entity, but without the strict grid layout. These are called amorphous solids, and they’re surprisingly common. Think of glass, rubber, plastic, and even tar.
In amorphous solids, the particles are still held relatively close together, and they’re not moving around freely. But instead of a repeating, ordered pattern, they’re arranged in a more random, disordered way. Imagine trying to pack marbles into a box without any specific order – you'll fill the space, but the marbles won't be in neat rows and columns. It’s like a crowd of people at a concert, all close together, but not in formation.

The lack of a long-range order in amorphous solids has some interesting consequences. For instance, glass, a classic amorphous solid, doesn't melt at a sharp, defined temperature like crystalline solids do. Instead, as it heats up, it gradually softens and becomes more viscous. This is why glassblowers can heat a piece of glass until it’s malleable enough to shape into intricate forms – it's transitioning through a semi-solid state rather than a clean break from solid to liquid.
Cultural Connection: Think of stained glass windows in cathedrals. The glass itself is amorphous, allowing light to pass through and refract in beautiful ways. The artists meticulously cut and shape these amorphous materials to create their masterpieces. It's a beautiful fusion of material science and art, where the "disorder" of the glass is harnessed for stunning effect.
Plastics are another fantastic example. Their long, chain-like molecules can be arranged in various ways, some more ordered than others. This flexibility in their structure is what makes them so versatile, allowing us to mold them into everything from sturdy car parts to flexible food packaging. The way these long polymer chains are tangled and packed determines whether the plastic is rigid or flexible.
Practical Tip: The "It'll Bounce Back" Rule
Have you ever dropped something made of plastic or rubber and it bounced? That's a characteristic of some amorphous solids. The disordered arrangement of their molecules allows them to deform under stress and then spring back to their original shape, unlike the more brittle, ordered crystalline solids which tend to fracture.
The Space Between the Atoms: Intermolecular Forces
So, what’s holding all these particles together in the first place? It’s a mix of forces, depending on the type of solid. In crystalline solids, as we’ve touched upon, we have strong ionic bonds, metallic bonds, and covalent bonds holding atoms together within a larger structure. But even in molecular crystals, where individual molecules are intact, there are weaker forces between these molecules. These are called intermolecular forces.
These intermolecular forces are like the invisible strings that keep our dance partners from drifting too far apart. They might not be as strong as the bonds within a molecule (like the covalent bonds holding oxygen and hydrogen together in water), but they are crucial for maintaining the solid state. Think of them as the friendly nods and polite conversation that keep a group of acquaintances together at a party.
The strength of these intermolecular forces directly impacts the melting point of a substance. Solids with stronger intermolecular forces require more energy (heat) to overcome these attractions and break free into a liquid state. This is why diamond, with its incredibly strong covalent bonds, has an astronomically high melting point, while something like dry ice (solid CO₂) sublimes (turns directly into gas) at a relatively low temperature due to weaker intermolecular forces.
Fun Fact: The "Sticky" Nature of Solids
Some solids, like certain adhesives or even very thick honey (which is technically a supercooled liquid, but behaves like a solid for practical purposes!), exhibit strong intermolecular forces or, in the case of adhesives, chemical bonding that makes them incredibly sticky. This "stickiness" is a direct result of the attractive forces between the particles of the solid and the surface it adheres to.
The Big Picture: Order and Function
Whether it’s the rigid, predictable structure of a crystal or the more flexible, disordered arrangement of an amorphous solid, the way particles are packed is fundamental to a material's properties. It dictates whether something is hard or soft, brittle or flexible, a conductor or an insulator.
Our world is built on these arrangements. The silicon chips in our computers rely on the precise crystalline structure of silicon. The resilience of tires comes from the tangled polymer chains in rubber. The beauty of a gemstone is a direct result of its ordered lattice. Even the very air we breathe, when frozen, forms delicate ice crystals with a specific, beautiful arrangement.
Understanding this microscopic world of particle arrangement isn't just for scientists in lab coats. It helps us appreciate the materials we use every day, how they work, and why they behave the way they do. It’s the hidden architecture that makes our tangible reality possible.
Reflection:
So, the next time you pick up a solid object – your phone, a book, a pebble from your garden – take a moment to consider the invisible dance happening within. It’s a universe of particles, each in its designated place, vibrating with quiet energy. Whether it's the perfect order of a crystal or the charming disarray of an amorphous solid, it’s this arrangement that gives it its form, its function, and its very existence. It’s a reminder that even in the most solid and stable things, there's a dynamic, ordered world at play, a testament to the fundamental principles that shape our universe, one particle at a time.
