Difference Between Endothermic And Exothermic Chemical Reactions
/endothermic-and-exothermic-reactions-602105_final-c4fdc462eb654ed09b542da86fd447e2.png)
Have you ever felt a chill run down your spine, or maybe a sudden warmth spreading through your hands? It turns out, the same magic that makes your hands tingle in the cold or a cozy fireplace feel so good is happening in the tiny world of chemical reactions. We're not talking about potions and spells here, but about the everyday dance of molecules that can either grab energy or let it go. It's a bit like a chemical "give and take."
Think of it like this: some chemical reactions are like super-powered energy hoarders. They need a big gulp of energy from their surroundings to even get started. Imagine trying to start a really stubborn campfire; you need a lot of initial effort, maybe a good puff of air or some dry tinder. These are our endothermic reactions, and they love to suck up the good stuff.
On the flip side, you have the generous souls of the chemical world. These are the exothermic reactions, and they're like little energy givers. They don't just get by; they actually release energy as they do their thing. Think of a sparkler on a birthday cake – poof! Light and heat just come bursting out.
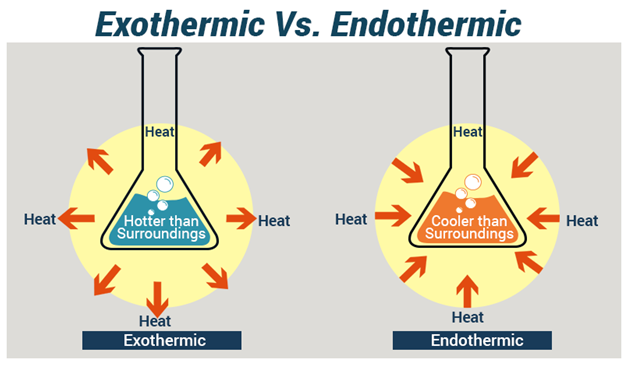
Let's dive a little deeper into our energy hoarders, the endothermic reactions. Their name kind of gives it away, right? "Endo" means "within," and "thermic" refers to heat. So, they're all about bringing heat into themselves. They're like a hungry plant reaching for sunlight.
A classic example, and one you might have experienced without even realizing it, is when you use a cold pack. You know, the kind you whack and it gets frosty? That's an endothermic reaction in action! The chemicals inside are grabbing heat from your sore muscle, making the pack cold and providing sweet relief. It's a little chemical magician making you feel better.
Another surprisingly endothermic friend is when you dissolve certain salts in water. Ever tried to make a really cold lemonade and noticed the glass felt cooler? That's a hint! Some dissolutions are endothermic, taking in a bit of that warmth from the liquid. It's like a tiny, refreshing ice bath for your drink.
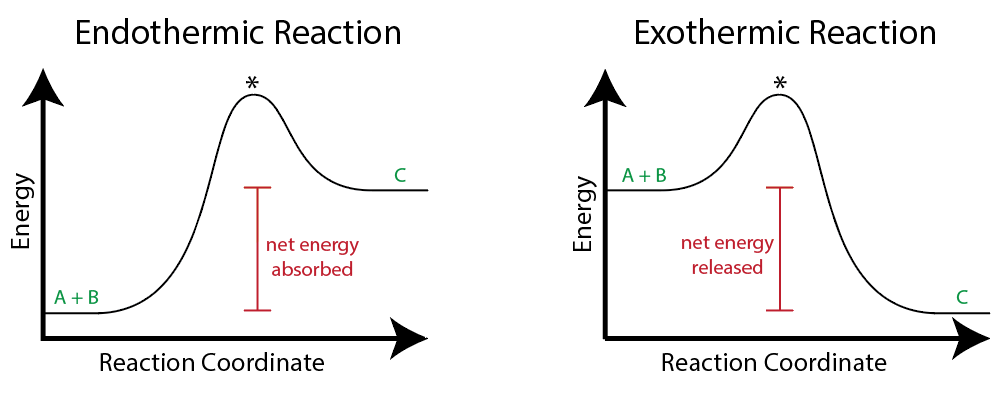
These endothermic reactions need that energy boost to break old chemical bonds and form new ones. They’re a bit like a builder needing bricks and mortar before they can construct something amazing. Without that energy input, they just can’t get the job done. It's a necessary step in their chemical construction project.
Now, let's swing over to the other side of the energy coin: the exothermic reactions. These are the show-offs, the ones that love to share their energy. "Exo" means "out," and again, "thermic" is heat. So, these reactions are all about sending heat out. They’re the life of the chemical party!
The most obvious and perhaps heartwarming example is combustion. Think of a roaring campfire, the kind you gather around on a chilly evening. That warmth radiating outwards? That's exothermic! The wood is burning, releasing all that stored energy as heat and light. It's nature's way of giving us a cozy hug.
And what about those instant hand warmers you can buy in the winter? You crack them open, give them a little shake, and voilà – they get toasty warm. Yep, you guessed it! Those are exothermic reactions working their magic, turning something simple into a little pocket of sunshine for your freezing fingers.
Even something as simple as rusting, which can be a bit annoying, is also exothermic! While it's a slow process, the formation of rust releases a tiny amount of heat. It's a gentle reminder that even mundane processes have an energetic story to tell. It’s a quiet release, unlike the dramatic fire.
When exothermic reactions happen, they're essentially releasing energy that was stored in the chemical bonds of the reactants. It’s like unwrapping a present that's been tightly packed; the energy that held it together is now free to roam. This energy often shows up as heat, but it can also be in the form of light or even sound.

So, what’s the big deal, you ask? Well, understanding this difference helps us do all sorts of cool things. We use exothermic reactions for power generation, from the fuel in our cars to the explosions that help build our cities. It's the driving force behind so much of our modern life.
And endothermic reactions? They're important too! They allow us to create those handy cold packs, and understanding them is crucial for things like refrigeration and air conditioning. Imagine a world without a cool escape from the summer heat – endothermic reactions make that possible! They are the unsung heroes of our comfort.
It’s like a constant balancing act in the universe. Some things need to take in energy to get going (endothermic), while others happily give it away (exothermic). It’s this perpetual exchange that keeps the chemical world – and by extension, our world – alive and kicking. It's a dance of energy, constantly moving and transforming.

Think about baking a cake. When you mix the ingredients, some interactions might be slightly endothermic, making the batter a tiny bit cooler. But then, when you bake it, the heat of the oven fuels a whole bunch of exothermic reactions that cause the batter to rise, set, and become that delicious treat. It's a culinary symphony of energy changes!
So, next time you feel a sudden chill or a welcome warmth, remember the amazing chemical reactions happening all around you. They’re not just abstract scientific concepts; they're the invisible forces that cook our food, keep us cool, and even provide the light from a birthday candle. They are the silent, energetic heartbeat of our everyday experiences.
It’s a beautiful reminder that even the most complex phenomena can be understood through simple, relatable analogies. From the sting of a cold pack to the glow of a fire, the difference between endothermic and exothermic reactions is a story of energy's generous spirit and its occasional embrace. They are the fundamental building blocks of change.
The world is full of these chemical wizards, some who take and some who give. And understanding their secrets simply adds another layer of wonder to the ordinary. It’s a fascinating glimpse into the energetic ballet that shapes our universe, one reaction at a time. They are the unsung heroes of the material world.
