Distinguish Between Physical Change And Chemical Change

Hey there, curious minds! Ever found yourself wondering about the little transformations happening all around us, from the toast popping up in the morning to that sparkly new science experiment you saw online? It turns out, there's a super cool way scientists like to sort these changes into two main camps: physical changes and chemical changes. Sounds a bit sciency, right? But trust me, it's way more like observing the world with a magnifying glass and saying, "Whoa, what's going on here?"
Let's dive in, no need to pull out your old textbooks or anything. Think of it like this: you're at a party, and things are happening. Some are just, you know, shifting around, while others are creating something totally new. That's kind of the vibe we're going for.
The "Same Stuff, Different Look" Club: Physical Changes
First up, let's chat about physical changes. These are the ones where a substance might look different, feel different, or even be in a different state (like solid, liquid, or gas), but it's still, at its core, the very same stuff. No new ingredients are being cooked up here!
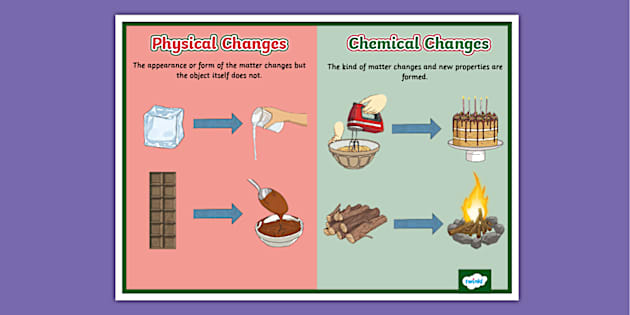
Imagine you have a piece of paper. You can rip it into a million tiny pieces, right? Does it stop being paper? Nope! It's still paper, just in a bunch of smaller bits. That's a perfect example of a physical change. You've altered its shape and size, but the fundamental composition remains the same. It's like rearranging your LEGO bricks – they're still LEGO bricks, just in a new formation.
Think about water. You can freeze it into ice (solid). You can melt that ice into liquid water. You can even boil that water and watch it turn into steam (gas). In all these instances, it's still H₂O. It's just showing off its different outfits! This is super common and happens all the time. Melting, freezing, boiling, condensing – these are all classic physical changes. They're reversible, too. You can melt ice back into water, and you can re-freeze water into ice. Easy peasy.

What about chopping up a carrot? You've got a whole carrot, and then you've got carrot slices. Still a carrot, just in a more manageable form for your salad. Or maybe you're folding a t-shirt. It goes from a flat pile to a neat rectangle. Still the same t-shirt, just tidier. These are all about changing the form or appearance, not the actual material itself. It's like putting on a costume; you're still you underneath!
The key thing to remember with physical changes is that no new substances are formed. The molecules that make up the substance are just moving around or arranged differently. It's like shuffling a deck of cards; the cards themselves haven't changed, just their order. Pretty neat, huh?
The "Surprise Ingredient!" Club: Chemical Changes
Now, let's venture into the exciting world of chemical changes. These are the ones where things get a little more adventurous. Here, a substance actually transforms into something entirely new. The molecules themselves rearrange and create different kinds of molecules. It's like a magical culinary experiment where you start with flour, eggs, and sugar, and end up with a delicious cake! The original ingredients are gone, replaced by something with different properties.

The most classic example? Burning wood. You start with a log of wood, and what do you end up with? Ash, smoke, and heat. The wood has fundamentally changed. It's not wood anymore. It's undergone a chemical reaction. You can't un-burn the wood and get your log back, right? That's a big clue that a chemical change has occurred. These are usually irreversible.
Think about baking a cake. You mix your batter, and it's a liquid. You bake it, and suddenly you have a solid, delicious cake. The chemical reactions happening in the oven have transformed the batter into something completely different. The eggs, flour, and sugar have combined and changed in ways that can't be undone to get your original ingredients back.
Rusting is another great one. You see a shiny new metal bike, and over time, if it's exposed to moisture and air, it develops that reddish-brown rust. The metal has reacted with the oxygen in the air to form iron oxide. It's a new substance with different properties – it's brittle and flaky, unlike the original metal. It's like your bike got a new, unwanted accessory!
What about when you digest your food? That's a massive series of chemical changes happening inside you! Your body breaks down complex molecules into simpler ones that your body can use. It's a whole biological chemical factory working away. Pretty amazing stuff!
So, how can you tell if a chemical change has happened? Keep an eye out for clues! Things like:
- Color changes: Like the rusting of iron or the browning of a banana peel.
- Gas production (bubbles!): Think of baking soda reacting with vinegar, which causes fizzing.
- Formation of a precipitate (a solid that forms in a liquid): Like when you mix two clear liquids and a cloudy solid appears.
- Temperature changes: Some reactions give off heat (exothermic), and others absorb heat (endothermic), making things feel hotter or colder.
- Light or sound production: Like a firework, which involves rapid chemical reactions.
These are all signals that something new is being created. It's like a secret code the universe is using to tell you, "Hey, a transformation is happening!"

Why Does It Even Matter?
You might be thinking, "Okay, cool, but why should I care?" Well, understanding the difference between physical and chemical changes helps us make sense of the world. It's the basis of cooking, cleaning, medicine, manufacturing, and so much more! When you're trying to figure out how to remove a stain (chemical change involved!) or how to make a perfect cup of tea (mostly physical changes, but the brewing is a bit of both!), knowing these concepts can be super helpful.
It's also just plain fascinating! It’s like being a detective, observing the world and figuring out the 'why' and 'how' behind all the transformations. The next time you see something change, take a moment. Ask yourself: Is it just changing its appearance, or is it becoming something entirely new? The answer might be simpler, and cooler, than you think!
So, next time you're in the kitchen, or looking at the changing seasons, or even just watching a candle burn, remember our two clubs: the "Same Stuff, Different Look" physical changes, and the "Surprise Ingredient!" chemical changes. They’re happening everywhere, all the time, making our world dynamic and endlessly interesting. Keep that curiosity alive!
