Does H2 Pd Reduce Esters
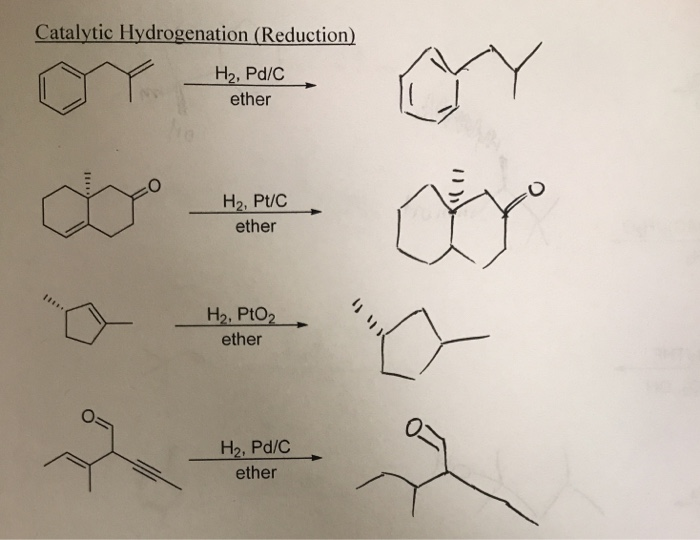
Hey there, science nerds and curious cats! Let's dive into something that might sound a little intimidating, but trust me, it's actually pretty cool. We're talking about esters and this fancy-sounding thing: H2 Pd. Sounds like a superhero team-up, right? H2 and Pd, fighting the good fight against… well, we'll get to that!
So, what even are esters? Think of them as the little scent machines of the world. They're responsible for all sorts of yummy smells. That banana smell? Yep, ester. Pineapple? Another ester. Even the sweet, floral notes in your favorite perfume? You guessed it – esters at work!
They're basically molecules that come from acids and alcohols. Imagine them as a cute couple, joined at the hip. The acid part and the alcohol part decided to get together and form something new. And the result? A molecule that smells delightful. Nature's little perfumers!
Now, what about this H2 Pd? This is where things get exciting. H2 is just hydrogen gas. You know, the stuff that makes balloons float and is super abundant. And Pd? That's palladium. It's a precious metal, shiny and a bit of a diva in the chemistry world. It likes to be in the spotlight and help out with reactions.
When you put them together, H2 and Pd, they often form a dynamic duo for a process called reduction. Think of reduction as taking something and making it a little bit simpler, or maybe changing its form in a significant way. It's like taking a complex Lego castle and breaking it down into its basic bricks.
So, the big question: does this superhero duo, H2 Pd, actually reduce esters? Can they change those scent-making molecules?
The answer is a resounding… sometimes!
It's not a simple yes or no. Chemistry is rarely that straightforward, is it? It's more like, "Well, it depends on the vibe."
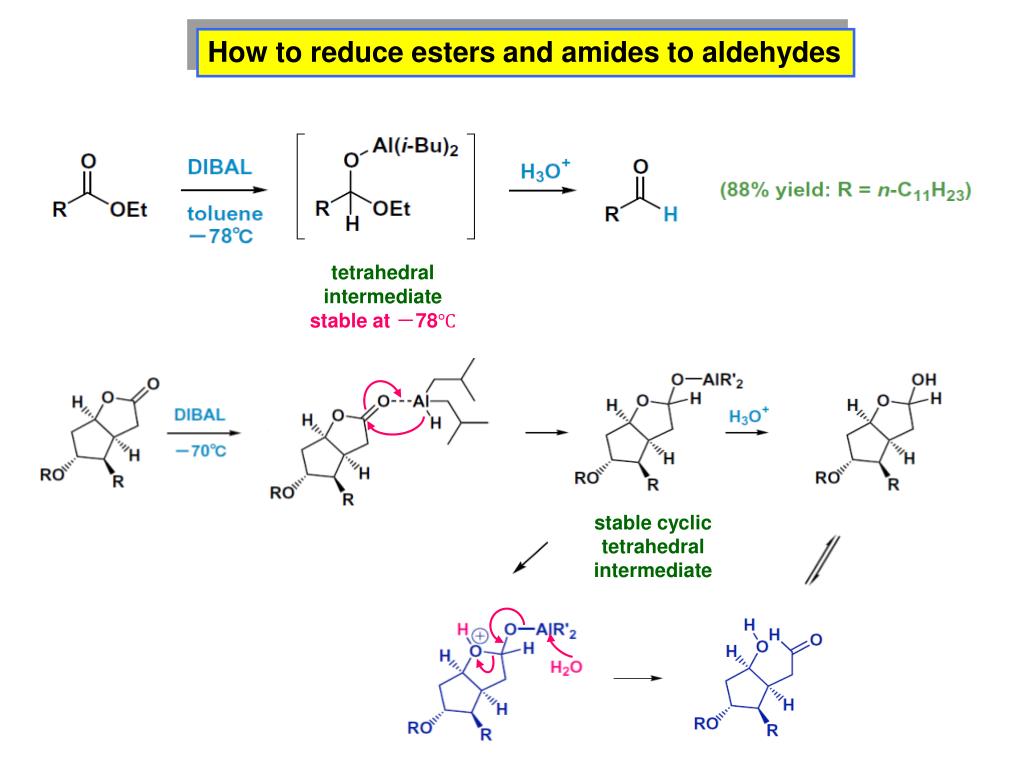
When H2 Pd gets together with an ester, it can perform a reduction. But what does that reduction look like? For an ester, it's usually a pretty drastic makeover. We're talking about breaking apart that ester bond. Remember the happy couple we talked about? Sometimes, H2 Pd comes along and says, "Hey, it's time for a divorce!"
The ester molecule gets split back into its original acid and alcohol components. It's like un-making the perfume to get the individual fragrant ingredients back. Pretty neat, huh?
However, there are a few catches. It's not like you can just toss any ester into a vat of H2 Pd and expect a perfect split. The conditions need to be just right.
Think of it like baking. You can't just throw flour and eggs into the oven and expect a cake. You need the right temperature, the right amount of time, and maybe some sugar and butter!
For H2 Pd to effectively reduce an ester, you usually need specific conditions. This often involves a solvent – that's like the liquid that helps everything mix and mingle. And the pressure of the hydrogen gas can matter. Sometimes you need a bit more oomph!
The type of ester itself plays a role too. Some esters are more "stubborn" than others. They're like the ones who really don't want that divorce. They need a bit more persuasion from H2 Pd to break apart.
But here's a super cool quirk: sometimes, H2 Pd is too good. It's like an overzealous chef who burns the cake. If you're not careful, H2 Pd can go beyond just splitting the ester.

It can actually reduce the alcohol that's formed from the split even further! And sometimes, it can even mess with other parts of the molecule if they're also susceptible to reduction. It's like, "Oops, did I also chop off the decorations from the cake?"
This is why chemists often use a catalyst. Remember how palladium likes to be in the spotlight? It acts as a catalyst. A catalyst is like a helpful friend who speeds up a reaction or makes it happen under milder conditions, without being used up themselves. Palladium is a real MVP in hydrogenation reactions like this.
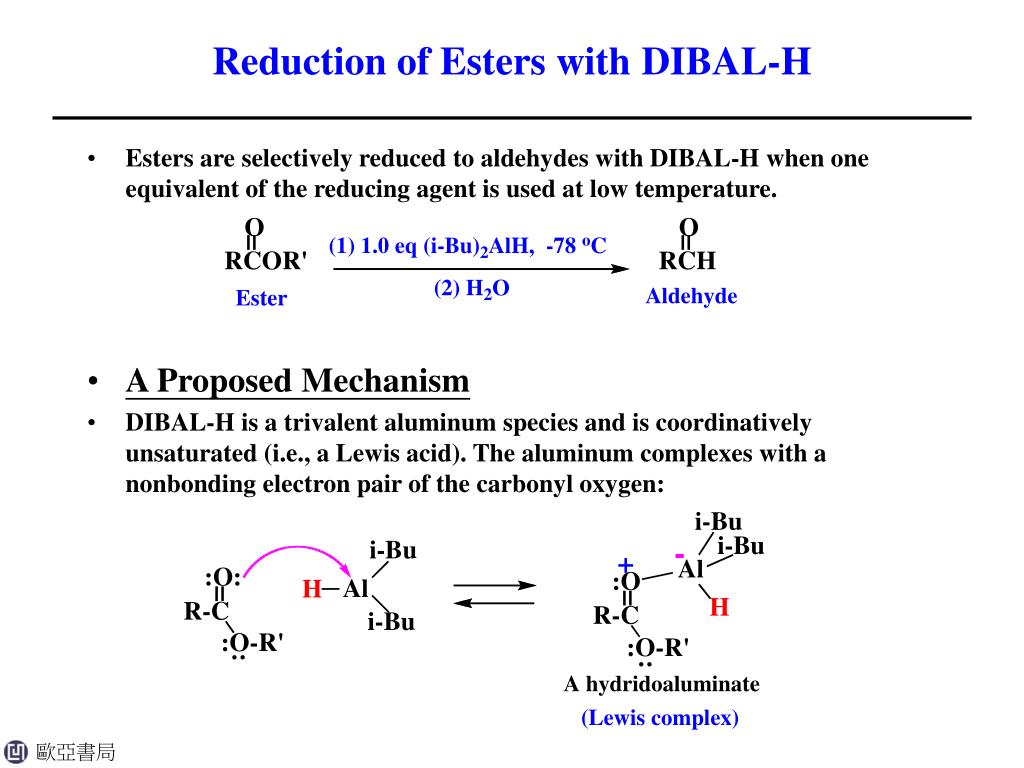
There are also other ways to reduce esters, which makes this whole topic even more fun. You can use different reducing agents. Think of it as having a toolbox full of different tools. Sometimes you need a hammer, sometimes you need a screwdriver. For esters, sometimes H2 Pd is the right tool, and sometimes you need something else, like lithium aluminum hydride (LiAlH4) – which sounds way scarier than it is, but it's a different kind of powerful reducer!
So, why is this even interesting to chat about? Well, it's about understanding how we can take molecules apart and put them back together. It's fundamental to making all sorts of things we use every day.

Think about the pharmaceutical industry. They need to create complex molecules for medicines. Understanding how to manipulate functional groups like esters is crucial. It's like knowing how to assemble and disassemble the most intricate puzzle.
Or think about the fragrance industry. If you want to create a new perfume, you might need to synthesize specific ester molecules. Knowing how to break them down or modify them is key to unlocking new scent profiles.
And honestly, there's just something inherently fascinating about these tiny building blocks of matter and how we can control their transformations. It’s like playing with molecular Lego, but with the power to make things smell amazing or create life-saving drugs!
So, the next time you smell a delicious fruit or a beautiful flower, remember the chemistry behind it. Remember the potential of a simple molecule and the powerful duo of H2 Pd. It’s a reminder that even in the seemingly complex world of chemistry, there are fun stories and unexpected transformations happening all the time. And yes, H2 Pd can reduce esters, but like any good story, it has its nuances, its challenges, and its moments of unexpected brilliance!
