How Does Surface Area Affect The Rate Of Diffusion

Hey there, science enthusiasts and curious minds! Ever wondered why your favorite tea bag seems to infuse its flavor so quickly into hot water, or why a tiny drop of food coloring can spread through a whole glass of water? It’s all thanks to a fascinating scientific principle called diffusion! It’s not just a lab concept; diffusion plays a vital role in so many aspects of our everyday lives, from the way our bodies work to how we enjoy our favorite foods and beverages.
Think about it: diffusion is essentially the natural tendency for molecules to spread out from an area where they are concentrated to an area where they are less concentrated. It’s like a gentle nudge that gets things moving, ensuring that substances mix and distribute themselves evenly. This process is super important for life itself! Our bodies rely on diffusion for everything from oxygen getting into our bloodstreams to nutrients reaching our cells. Without it, we simply wouldn't function!
Beyond our biology, diffusion is at play in countless practical scenarios. That lovely aroma of freshly baked cookies wafting through your house? Diffusion of scent molecules. Making a soothing cup of chamomile tea? Diffusion of flavor and beneficial compounds from the tea leaves. Even the way your laundry detergent disperses evenly in the washing machine is a testament to diffusion. And let's not forget about the simple pleasure of a perfectly infused drink, where the colors and flavors meld together beautifully.
But have you ever noticed that sometimes diffusion seems to happen faster than other times? The secret ingredient here is often surface area! Imagine trying to dissolve a giant cube of sugar versus using granulated sugar. The granulated sugar, with its many tiny grains, has a much larger total surface exposed to the water. This means more points of contact for the water molecules to interact with the sugar molecules, speeding up the dissolving process. The same principle applies to diffusion in general.
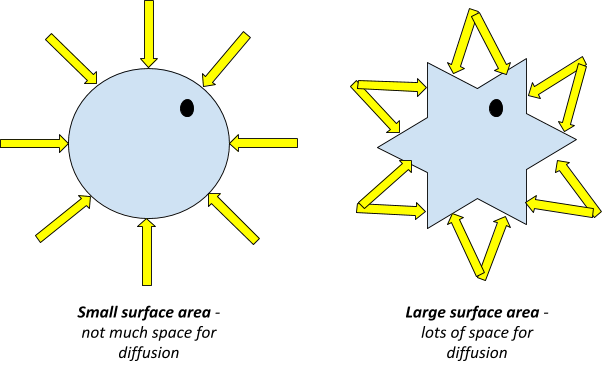
So, how does surface area affect the rate of diffusion? It’s quite straightforward: the greater the surface area, the faster the diffusion will occur. This is because diffusion happens at the boundary between two substances. When you increase the surface area, you’re essentially increasing the amount of space where these molecules can interact and move past each other. Think of it like having more doors for molecules to enter and exit a particular space. More doors, more traffic, and faster movement!
This principle is why finely chopped vegetables cook faster than whole ones, or why a teabag is designed with a large, porous surface. To enjoy the benefits of faster diffusion in your own life, consider these practical tips. When brewing tea or coffee, using finer grounds or tea leaves will lead to a more robust and quicker infusion. If you're cooking, chopping ingredients into smaller pieces will significantly reduce cooking times. Even when you're trying to cool down a drink, adding ice cubes offers a larger surface area for heat transfer, making it chill faster. So, the next time you're enjoying a warm beverage or a delicious meal, take a moment to appreciate the invisible dance of molecules and how a little thing like surface area can make a big difference!
