How Does The Temperature Affect The Rate Of Diffusion
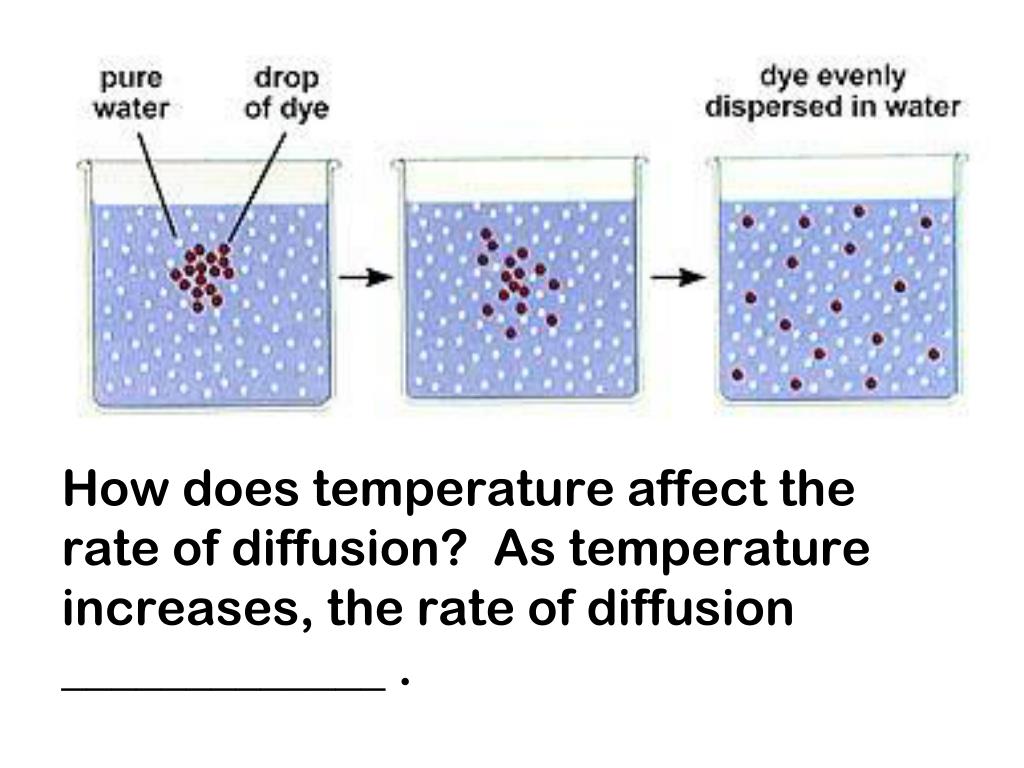
Hey there! Grab your mug, settle in, because we're about to spill the beans on something super cool: how temperature messes with diffusion. Yeah, diffusion. You know, that thing where stuff just… moves around on its own? Like when you spray perfume, and suddenly the whole room smells amazing. Or when you drop a tea bag in hot water, and boom! Instant tea. Pretty neat, right?
So, what’s the deal with temperature? Turns out, it’s a huge player. Think of it like this: are your molecules chilling out, or are they having a massive rave? Temperature tells them which party they’re at.
The Speedy World of Hot Molecules
When things get warm, it’s like everyone just chugged a triple espresso. Molecules go absolutely bonkers! They start vibrating, jiggling, and bumping into each other like kids on a sugar high at a bouncy castle. This energetic dance is what makes diffusion happen faster. More movement, more collisions, more spreading out. It’s like a microscopic mosh pit, but way more organized (mostly).
Imagine dropping a tiny speck of food coloring into a glass of ice-cold water. What happens? It just kinda sits there, right? Then, you pour the same speck into hot water. BAM! Swirling colors, spreading out like wildfire. That’s temperature showing off its power. It’s the ultimate party starter for molecules.
This isn’t just about pretty colors, though. This speed-up is happening all the time, in all sorts of things. It’s pretty fundamental to how the world works, which is kind of mind-blowing if you think about it too hard. But don’t worry, we’re just chatting here!
Why All the Jiggling?
Okay, so why does heat make things jiggle more? It’s all about energy. Heat is basically just a measure of how much kinetic energy the molecules in a substance have. Kinetic energy, in super simple terms, is the energy of motion. So, more heat means more energy, which means more motion. Simple as that, really.
Think of a quiet room versus a concert hall during a rock anthem. In the quiet room, people are just milling around, maybe having a quiet chat. In the concert hall, they’re jumping, dancing, and probably bumping into each other a lot. The concert hall is the high-temperature environment, and the quiet room is the low-temperature one. See the connection?
This increased jiggling means molecules are more likely to break away from their neighbors and mingle with others. They’re less likely to be stuck in place. It’s like they’re all trying to escape the dance floor to go get some snacks, but on a molecular level. And because they’re moving so much, they spread out faster, filling up any available space.

When Things Cool Down: The Slow Lane
Now, let’s flip the script. What happens when things get chilly? Suddenly, it’s like someone’s turned down the music, and everyone’s deciding to take a long, slow nap. Molecules lose energy. They slow down. The energetic rave turns into a sleepy pajama party.
Diffusion in cold temperatures is like watching paint dry. Seriously. It’s slow. Molecules are less inclined to move, less likely to bump into each other with any real force. They’re more content to just hang out where they are, being all chill and mellow. Not exactly a recipe for rapid spreading, is it?
Ever put a steak in the freezer? It doesn’t really defrost instantly, does it? And if you were to try and infuse flavor into something frozen, good luck! The molecules responsible for flavor are just too sluggish. They’re not in the mood for a speedy migration. This is the power of cold – it puts the brakes on diffusion.
So, next time you’re waiting for something to dissolve in a cold drink, just remember: you’re witnessing the molecular equivalent of a glacial pace. It’s science, baby, but it can feel like torture sometimes, can’t it?
The Science of Slowness
Why do they slow down? It’s the same reason they sped up: energy. When the temperature drops, molecules have less kinetic energy. Less energy means less movement. It’s like trying to run a marathon after a massive Thanksgiving dinner – you’re just not going to have the get-up-and-go.

They still move, of course. Even at absolute zero (which is basically impossible to reach, but hey, science!), there's still a tiny bit of motion. But at typical cold temperatures, the movement is significantly reduced. They’re not exactly sprinting; they’re more like… shuffling. Very, very slowly.
This reduced movement means fewer collisions. And fewer collisions mean less chance of molecules bouncing off each other and spreading into new areas. They’re more likely to just stay put, maybe nudge their neighbor a little, but not go on any grand adventures. It’s the molecular equivalent of social distancing, but they’re not even trying!
It’s All About the Kinetic Energy, Folks!
So, the big takeaway here, the absolute golden nugget of this whole chat, is that kinetic energy is the name of the game. Temperature is just the dial that controls how much kinetic energy our little molecular friends have. Hotter temperature = more kinetic energy = faster diffusion. Colder temperature = less kinetic energy = slower diffusion.
Think about it. Your body is a pretty warm place, right? That’s why so many chemical reactions can happen inside you at a decent speed. If your body temperature suddenly plummeted, a lot of those essential processes would grind to a halt. Pretty dramatic, huh?
And it’s not just about liquids and gases. Even in solids, where molecules are packed in super tight and don’t move around much, temperature still plays a role. Heating up a solid can make its atoms vibrate more, which can lead to subtle diffusion, like how metals can “age” and change their properties over time due to slow atomic movement. It’s mind-bogglingly cool!

A Little Analogy to Seal the Deal
Let’s try one more analogy to really hammer this home. Imagine a crowded room at a party.
If the music is pumping and everyone’s hyped (high temperature), people are bumping into each other, moving around, and generally spreading out to find more space or chat with different groups. Diffusion is rapid!
Now, imagine that same room, but the power’s gone out, it’s late, and people are tired and sleepy (low temperature). They’re not going to be doing much bumping or moving. They’ll probably just stay in their little huddles, barely shifting. Diffusion is practically non-existent.
See? It’s that simple. The amount of energy, the “liveliness” of the molecules, directly dictates how quickly they can spread out and mix. Temperature is the ultimate mood setter for our molecular parties.
Real-World Wonders of Temperature and Diffusion
So, where do we see this in action, besides the obvious perfume and tea examples? Oh, everywhere!
Think about cooking. When you bake a cake, the heat causes all the ingredients to diffuse and interact, creating that delicious final product. If you tried to mix a cake batter at room temperature and then just popped it in the oven, it wouldn’t be the same, would it? The heat is essential for the molecules to mingle and transform.
Or consider the human body. Our cells are constantly taking in nutrients and expelling waste products. This happens through diffusion across cell membranes. And guess what influences the speed of that diffusion? Yep, our body temperature!
Even in the natural world, it’s a big deal. Ocean currents and atmospheric circulation are, in part, driven by temperature differences and the resulting diffusion of heat and other substances. It’s a subtle but powerful force shaping our planet.
And what about things like medicine? When doctors administer injections, the medication needs to diffuse from the injection site into the surrounding tissues to do its job. The rate of this diffusion can be influenced by the temperature of the body. Pretty important stuff!
The Sweet Spot for Science
It’s fascinating to think about how these seemingly small molecular movements, influenced by something as simple as temperature, have such massive implications. It’s not just a theoretical concept; it’s the engine behind so many everyday phenomena and complex biological and environmental processes.
So, the next time you feel a bit chilly or a bit warm, just remember that you’re not just feeling a sensation. You’re also experiencing a fundamental aspect of how the universe works. You’re witnessing the molecular dance, sped up or slowed down by the magic of temperature. Pretty cool, right? Now, about that refill…?
