How To Calculate Rf Value In Paper Chromatography
Ever found yourself mesmerized by those colorful streaks separating on a piece of paper, wondering what magic is at play? Welcome to the wonderfully chill world of paper chromatography, a technique that’s as much about science as it is about a relaxed afternoon experiment. Think of it as a gentle journey for tiny molecules, a bit like a lazy river ride where each substance takes a unique path. And at the heart of understanding this journey is a simple yet super useful number: the Rf value.
Don't let the fancy name scare you. Calculating the Rf value is about as complicated as brewing your morning coffee – maybe even easier! It’s essentially a way to measure how far a substance traveled up the paper compared to how far the solvent (the liquid that carries everything along) traveled. It’s the scientific equivalent of saying, "Okay, so this guy went this far, and the river went that far. How did they stack up?"
Imagine you’re at a music festival, and you’ve got a bunch of your favorite bands on different stages. The Rf value is like ranking their popularity based on how many people managed to get to their stage before the next set started. Some bands might be absolute headliners, drawing massive crowds who rush to them (high Rf), while others might be the chill, acoustic acts playing in a quiet corner, attracting a smaller, more dedicated following who take their time getting there (lower Rf). It’s all about relative movement!
So, What Exactly is This Rf Value?
Alright, let’s get down to the nitty-gritty, but keepin’ it super light, of course. The Rf value stands for Retention Factor. Sounds a bit technical, right? But really, it’s just a ratio. Think of it as a fingerprint for a particular chemical compound under specific conditions. Every chemical, when placed on a paper with a certain solvent, will travel a predictable distance relative to the solvent front. That predictability is the Rf value.
Why is this so cool? Because it helps us identify unknown substances. If you’re trying to figure out what ink is in your favorite pen (a classic paper chromatography experiment, by the way!), or perhaps analyze the pigments in a plant leaf, the Rf value can be your guide. It’s like having a detective’s notebook for the microscopic world.
The beauty of it is that the Rf value is dimensionless. That means it doesn't have units like centimeters or inches. It’s just a number, usually between 0 and 1. A value of 0 means the substance didn't move at all, staying put right where you dropped it. A value of 1 means it zoomed all the way up with the solvent, practically hitchhiking a ride.
The Simple Formula: Your Scientific Sidekick
Ready for the formula? Deep breaths. It’s not going to bite. In fact, it’s quite friendly:
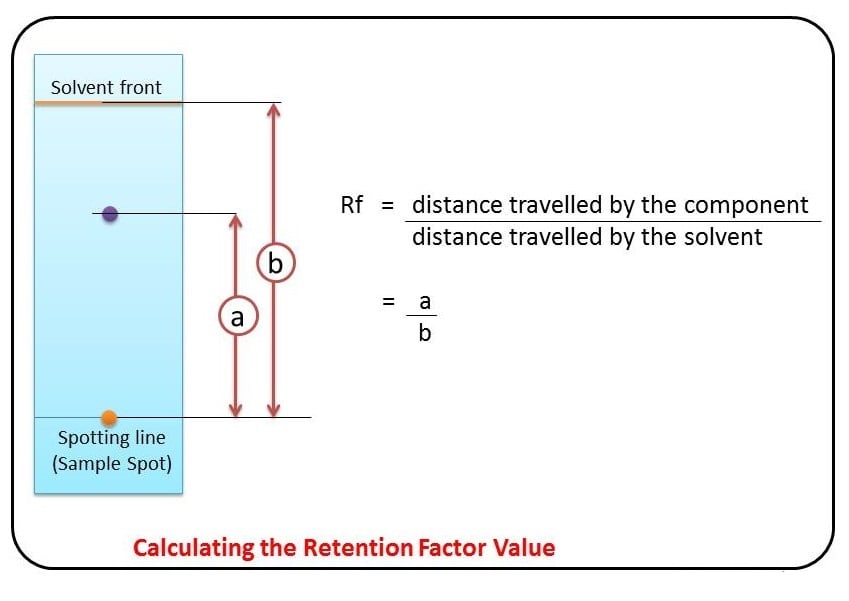
Rf = (Distance traveled by the substance) / (Distance traveled by the solvent front)
That’s it. Seriously. No calculus, no quadratic equations. Just a straightforward division. You measure how far your spot of interest has moved from its starting point, and then you measure how far the liquid has crept up the paper. Divide the first by the second, and voilà! You’ve got your Rf value.

Let’s break down what each part means:
- Distance traveled by the substance: This is the distance from the origin (where you initially spotted your sample) to the center of the separated spot (or the middle of the colored band if it’s spread out).
- Distance traveled by the solvent front: This is the distance from the origin to the leading edge of the solvent as it moves up the paper.
Think of it like this: You're running a race. The substance is you, and the solvent front is the finish line. Your Rf value is your pace relative to the overall race duration. Are you sprinting ahead, or are you strolling and enjoying the scenery?
Let's Get Practical: Your Step-by-Step Rf Guide
Now, how do you actually do this? It’s a fun, hands-on process, perfect for a rainy Saturday or a curious afternoon. Imagine you're a mad scientist, but, you know, a really chill one.
Step 1: Prepare Your Paper and Spot Your Sample
You’ll need a piece of chromatography paper. This isn't your average printer paper; it's specially designed with a uniform structure that allows for good separation. You can get these online or from science supply stores. Gently draw a horizontal line near the bottom of the paper with a pencil. This is your origin line. Then, carefully spot your sample (e.g., a tiny drop of ink) onto this line. Let it dry completely. If you're using multiple samples, space them out along the origin line.
Fun Fact: The type of paper matters! Different papers have different pore sizes and properties, which can affect the Rf values. So, if you're comparing results, make sure you're using the same paper.
Step 2: Set Up Your Developing Chamber
This is where the magic begins. You’ll need a container to hold your solvent, like a tall beaker or a jar. Make sure it’s tall enough so the paper can hang inside without touching the bottom. Pour a small amount of your chosen solvent into the chamber. The solvent is the mobile phase – the vehicle carrying your samples. Common solvents include water, ethanol, or mixtures of different liquids, depending on what you're trying to separate.

Important Tip: The solvent level should be below the origin line. If the solvent touches the origin line, your samples will just dissolve into the pool and won't travel up the paper. It's like starting a race with your feet already in the water – not ideal!
Step 3: Let the Separation Happen
Carefully place your paper into the developing chamber, usually by resting the top edge over a rod or clip, allowing the bottom edge to dip into the solvent. The solvent will start to move up the paper by capillary action – that’s the same force that pulls water up a plant's roots. As the solvent moves, it carries your sample components with it. Different components will dissolve in the solvent to different degrees and will interact differently with the paper, causing them to separate into distinct spots or bands.
This step requires patience. You might want to cover the chamber to prevent the solvent from evaporating too quickly. Grab a book, put on some chill tunes, and let nature do its thing. It’s a beautifully passive process.
Step 4: Mark the Solvent Front and the Spots
Once the solvent has traveled a good distance up the paper (but before it reaches the very top!), carefully remove the paper from the chamber. Immediately mark the solvent front with a pencil. This is the highest point the solvent reached. Then, locate the center of each separated spot (or the middle of each colored band) and mark its position on the paper. If your spots are faint, you might need to use a UV light if your compounds are UV-active, or a developing spray.
Pro-Tip: Drawing the origin line and solvent front with a pencil is key. Ink lines would just run with the solvent and mess up your results! Pencils use graphite, which is insoluble in most solvents used in chromatography.
Step 5: Measure and Calculate!
Now for the moment of truth! Using a ruler, measure the distance from the origin line to the center of each separated spot. Let's call this distance_substance. Then, measure the distance from the origin line to the solvent front. Let's call this distance_solvent.

Plug these numbers into our friendly formula:
Rf = distance_substance / distance_solvent
For example, if a spot traveled 6 cm and the solvent front traveled 10 cm, the Rf value would be 6 cm / 10 cm = 0.6. This value (0.6) is dimensionless and specific to that substance under those exact conditions (paper type, solvent, temperature).
Why is this Rf Value So Important? (Beyond the Cool Science Stuff)
Calculating Rf values is more than just a lab exercise. It’s a fundamental technique used in many fields:
- Drug Analysis: Identifying and quantifying active ingredients in pharmaceuticals.
- Forensics: Analyzing inks from documents, identifying dyes in textiles, or even analyzing trace evidence.
- Environmental Science: Detecting pollutants in water or soil.
- Food Industry: Checking for the presence of artificial colorings or preservatives.
- Art Conservation: Identifying pigments used in historical artworks.
Think of it like this: when you’re trying to find a specific ingredient in a recipe, you’re essentially looking for a unique combination of properties. Chromatography, and the Rf value, help us do that on a molecular level. It's a way of saying, "This particular molecule behaves this way, making it distinct from others."
Cultural Nod: Remember those vibrant, hand-dyed fabrics of India or the intricate patterns of Japanese kimonos? Chromatography can be used to analyze the natural dyes that create these beautiful textiles, helping us understand traditional dyeing techniques and preserve cultural heritage.

Keeping it Consistent: The Little Things That Matter
Since the Rf value is so specific to the conditions, it’s crucial to keep everything consistent if you want to compare results or identify substances accurately. What does this mean in practice?
- The Solvent: Always use the same solvent mixture. Even small changes in the ratio of liquids can alter the Rf values.
- The Paper: Use the same type and brand of chromatography paper.
- Temperature: Room temperature is usually fine, but significant temperature fluctuations can affect solvent evaporation and flow rate.
- Spotting Technique: Try to spot the same concentration and volume of sample.
- Development Time: Allow the solvent to run for a similar amount of time (or to a similar height) if comparing multiple runs.
It’s like when you’re trying to perfect your grandmother’s famous cookie recipe. You can’t just swap out the flour for almond flour one day and then wonder why the cookies taste different! Consistency is key to getting that perfect, repeatable result.
Fun Fact: The concept of chromatography was first developed by Russian botanist Mikhail Tsvet in the early 20th century. He used it to separate plant pigments like chlorophyll. So, in a way, we owe our understanding of plant colors to a very organized scientist!
A Reflection: The Journey of Separation in Daily Life
It's fascinating how this seemingly simple scientific principle can resonate with our everyday lives. We're all on our own personal journeys, aren't we? Each of us, like the components in chromatography, has unique properties that interact with the "solvent" of our environment – our experiences, relationships, and challenges.
Sometimes, we might feel like a substance that travels far and fast, easily adapting and moving with the flow. Other times, we might feel more anchored, our journey slower, our interactions with the world leading to more concentrated, defined experiences. The Rf value, in its essence, reminds us that movement and separation are natural processes. It’s about understanding our place, our speed, and our unique path through the world.
So, the next time you see those colorful bands on a paper, remember the Rf value. It’s not just a number; it’s a story of separation, a fingerprint of identity, and a gentle reminder that every substance, and every person, has their own distinct journey. And that, in itself, is a beautiful thing to understand.
