How To Find The Molecular Formula From Empirical

Imagine you're a detective, but instead of solving crimes, you're solving the secret makeup of tiny things – molecules! And sometimes, these molecules play a little hide-and-seek. They're like the shy celebrities of the science world, happy to show you their general vibe but keeping their exact, full glamorous entourage a secret. That's where we come in, ready to uncover their true identity!
You see, scientists often start by figuring out the simplest ratio of ingredients in a molecule. Think of it like this: if you're making a fruit salad and you know you're using apples, bananas, and strawberries, you might figure out that for every 2 apples, you use 1 banana and 3 strawberries. That's your empirical formula – the most basic, reduced version of the recipe. It tells you the kind of ingredients and their basic proportion, but not the actual quantity in the whole glorious salad.
For example, let's say we're talking about water. We know it's made of hydrogen and oxygen. The simplest ratio you can get is 2 hydrogens for every 1 oxygen. That's our empirical formula: H₂O. Easy peasy! But what if we're looking at something a little more complex, like sugar? The simplest ratio might be 1 carbon, 2 hydrogens, and 1 oxygen (CH₂O). Sounds simple enough, right? But is that really what a sugar molecule looks like in its full glory? Does a single sugar cube really only have one carbon atom? That would be like a party with only one guest! Not much fun.
This is where the magic happens, the part that makes you feel like you're in on a delicious secret. That CH₂O is just the blueprint, the most basic building block. The actual sugar molecule, the one that makes your cookies sweet and your lemonade delightful, is actually a lot bigger! It’s like finding out the simple recipe for a cookie is flour, sugar, and butter, but the actual cookie you're eating is a giant, triple-layer monstrosity of deliciousness. The empirical formula is just the idea of the ingredients, while the molecular formula is the full, actual, party-ready recipe.
So, how do we go from the shy, simple ratio to the grand, molecular party? We need a little extra clue. Imagine our detective needs a crucial piece of evidence. In the molecular world, this crucial piece of evidence is the molar mass of the molecule. This is basically the 'weight' of one whole, complete molecule. It’s like finding out how much your entire fruit salad actually weighs.

Think of our sugar example again. We know the empirical formula is CH₂O. Let's pretend we figure out the weight of one carbon atom is about 12 grams, one hydrogen atom is about 1 gram, and one oxygen atom is about 16 grams. So, the weight of our simple CH₂O unit would be 12 + (2 * 1) + 16 = 28 grams. This is the weight of the simplest possible ingredient combination.
Now, let's say our detective work, our super-accurate weighing of the actual, full molecule, tells us that a complete sugar molecule weighs a whopping 168 grams. Woah! That’s a lot more than our 28-gram building block. This is the moment of revelation! We realize that our simple CH₂O unit is not the whole story.
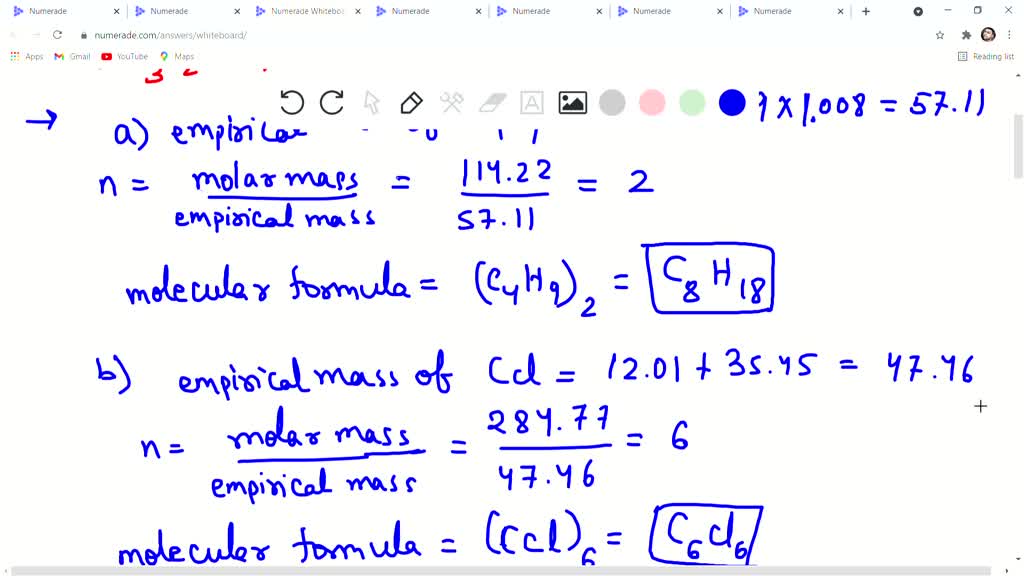
To find the full, glorious molecular formula, we just need to figure out how many of our simple building blocks fit into the grand, actual molecule. It’s like asking, "How many 28-gram units do I need to add up to get 168 grams?" We simply divide the big weight (168 grams) by the small weight (28 grams). 168 / 28 = 6. Bingo!
This number, 6, tells us that the actual sugar molecule is made up of 6 of those simple CH₂O units. So, we take our empirical formula (CH₂O) and multiply each of its little numbers by that magic '6'. So, C becomes C₆, H₂ becomes H₁₂, and O becomes O₆. And voilà! The molecular formula for our sugar is C₆H₁₂O₆. It’s like unlocking the full, delicious recipe!
It’s a beautiful dance between the simple and the complex. The empirical formula gives us the fundamental truth, the essential spirit of the molecule. The molecular formula then reveals its full, magnificent form, the actual entity that does all the wonderful things it does, whether it’s making our water life-giving or our sugar sweet. It’s a reminder that even the most complex and wonderful things can often be understood by starting with their simplest, most elegant foundation. And that, my friends, is a little bit of scientific magic we can all enjoy!
