Hydrogen Ions Are Attracted To Which Electrode During Electrolysis
Ever wondered what makes those cool science experiments work, the ones where electricity seems to pull things apart? It's a little bit like magic, but it's all thanks to some tiny, charged particles. Today, we're going to talk about one of the stars of the show during electrolysis: hydrogen ions. Understanding where they go during this process is super interesting and can unlock a whole world of science for you!
So, why is this even fun or useful? For starters, it's the key to understanding how we can split water into its basic components, hydrogen and oxygen. This has huge implications for clean energy and a whole lot more. For beginners, it's a fantastic way to grasp fundamental chemistry concepts without getting bogged down in jargon. Families can turn this into a captivating home experiment, sparking curiosity in younger minds. And for hobbyists, whether you're into DIY electronics or chemistry projects, knowing this principle opens doors to more advanced explorations.
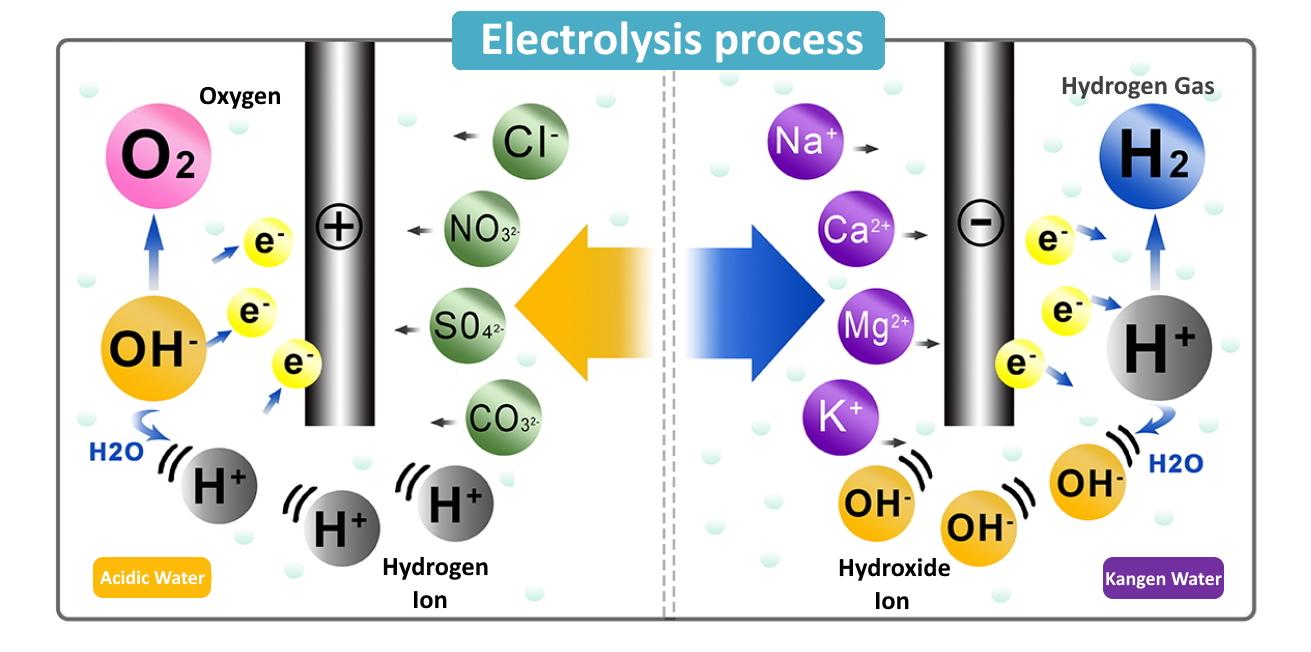
Let's get down to business. During electrolysis, we pass an electric current through a liquid, usually water with something dissolved in it to make it conductive. This liquid is called an electrolyte. This current creates two electrodes: one with a positive charge and one with a negative charge. Think of them like opposite magnets!
Now, about our hydrogen ions. These guys are atoms that have either lost or gained electrons, giving them an electric charge. Specifically, a hydrogen ion (often written as H⁺) has a positive charge. And what do positively charged things like to do when faced with electricity? They're attracted to the opposite charge! That means hydrogen ions are strongly attracted to the negative electrode.

The negative electrode is called the cathode. So, during electrolysis, you'll see hydrogen ions zipping towards the cathode. When they get there, they often pick up electrons and turn back into neutral hydrogen atoms, which can then form hydrogen gas (H₂). This is often seen as bubbles forming at the cathode. It's a simple yet powerful demonstration of electrical forces at work!
You might see this in action when you try to split water. You’ll typically observe bubbles forming more vigorously at one electrode than the other – that’s your hydrogen gas being produced at the cathode! Variations of electrolysis are used in everyday life, from electroplating jewelry to purifying metals. It's a foundational process with far-reaching applications.

Want to get started? You don't need a fancy lab! A simple experiment can be done with a 9-volt battery, some paperclips, water, and a pinch of salt or baking soda to make the water conductive. Remember to always have adult supervision for any experiments involving electricity or chemicals. Observing the bubbles is the most direct way to see where your hydrogen ions are headed – towards the negative pole!
So, the next time you hear about electrolysis, you'll know that those positively charged hydrogen ions are on a mission, heading straight for the cathode, the negative electrode. It’s a fascinating peek into the unseen world of atoms and electricity, and it’s surprisingly easy to appreciate once you know the secret!
