In A Fractionating Column What Process Is Caused By Heating
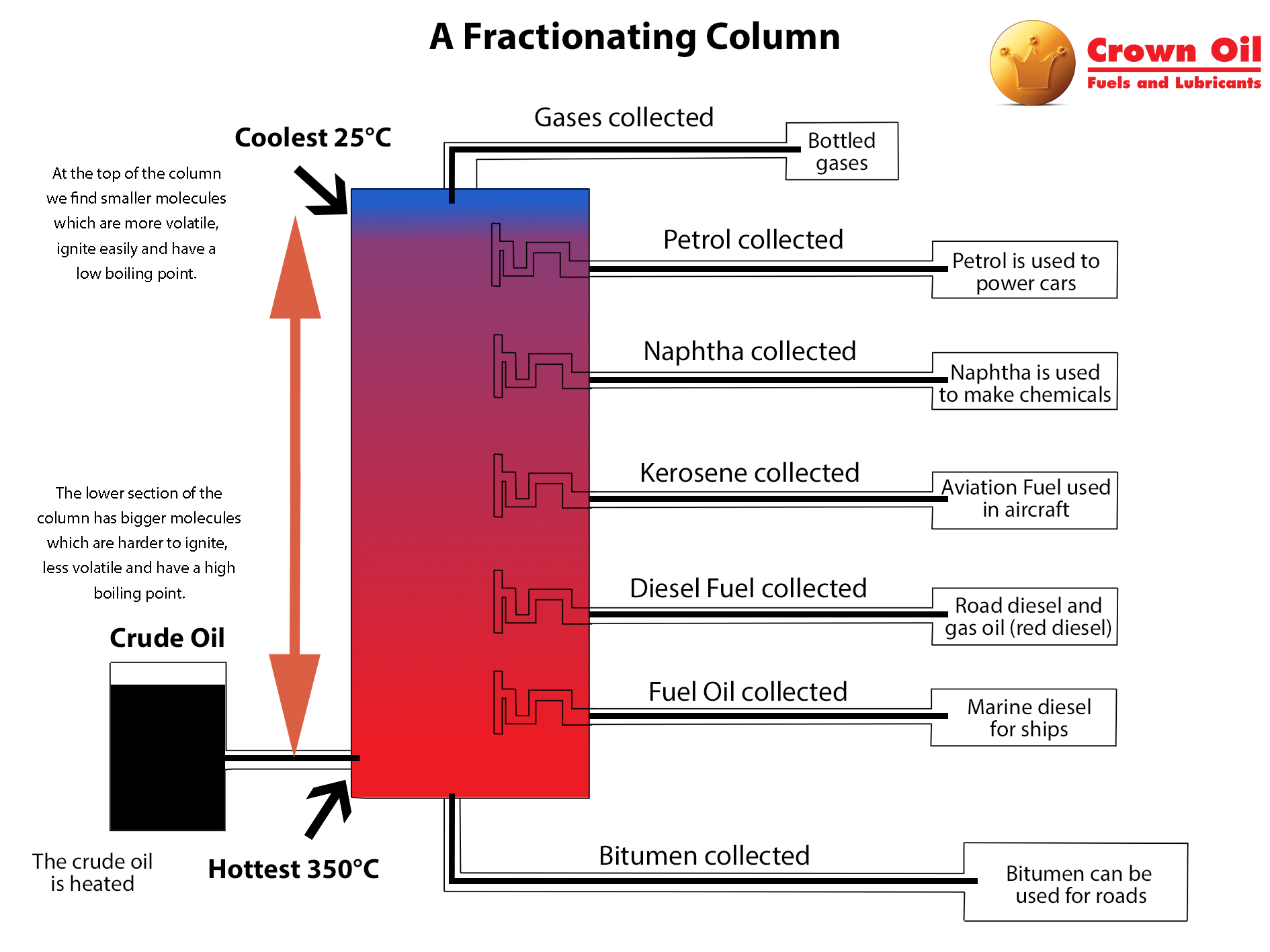
Ever wondered how we get so many different things from one source, like how crude oil turns into gasoline, plastics, and so many other useful products? Or maybe you've seen a science experiment that magically separates colored liquids? The magic behind a lot of these separations, both in the lab and in industry, is a fascinating process that happens in something called a fractionating column. It's a pretty cool concept, and understanding it isn't as complicated as it might sound. In fact, it's all about a simple, yet powerful, effect caused by heating.
So, what exactly happens when you apply heat inside a fractionating column? The core process at play is distillation, specifically fractional distillation. Imagine you have a mixture of liquids with different boiling points. When you heat this mixture, the liquid with the lowest boiling point will start to turn into a gas (or vapor) first. This vapor then rises up the column. As it rises, it encounters cooler temperatures. As the vapor cools, it condenses back into a liquid. The trick of the fractionating column is that it's designed with many "trays" or "plates" at different heights. Each tray is cooler than the one below it.
As the vapor rises and cools, it condenses on the trays. But here's where it gets interesting: as more vapor rises from below, it heats the liquid on the trays. This re-vaporizes the components with lower boiling points, causing them to rise further up the column. Meanwhile, the components with higher boiling points, which didn't vaporize as easily in the first place, tend to condense and drip back down. This continuous cycle of vaporization and condensation, happening at various levels within the column, effectively separates the mixture into its individual components, or fractions, based on their boiling points. The result is a much purer separation than you'd get from simple distillation.
The benefits are huge! In the oil refining industry, fractional distillation is absolutely essential. It allows us to take crude oil, a complex mix of hydrocarbons, and separate it into useful fuels like gasoline, diesel, kerosene, and even the raw materials for plastics. In the chemical industry, it's used to purify solvents and create specific chemical compounds. Even in something as everyday as producing alcoholic beverages, distillation plays a key role in concentrating the alcohol.
Looking for ways to explore this? In a school science lab, you might see simpler versions of distillation. For a fun, at-home experiment (with adult supervision, of course!), you could try separating colored water. While not true fractional distillation, you can see the principle of evaporation and condensation. A more relatable, though less direct, example is thinking about how steam from a boiling pot of water rises and then condenses on cooler surfaces like windows or cabinet doors. For a deeper dive, there are many fantastic educational videos online that animate the process beautifully. It's a testament to how fundamental principles of physics and chemistry shape so much of our modern world!
