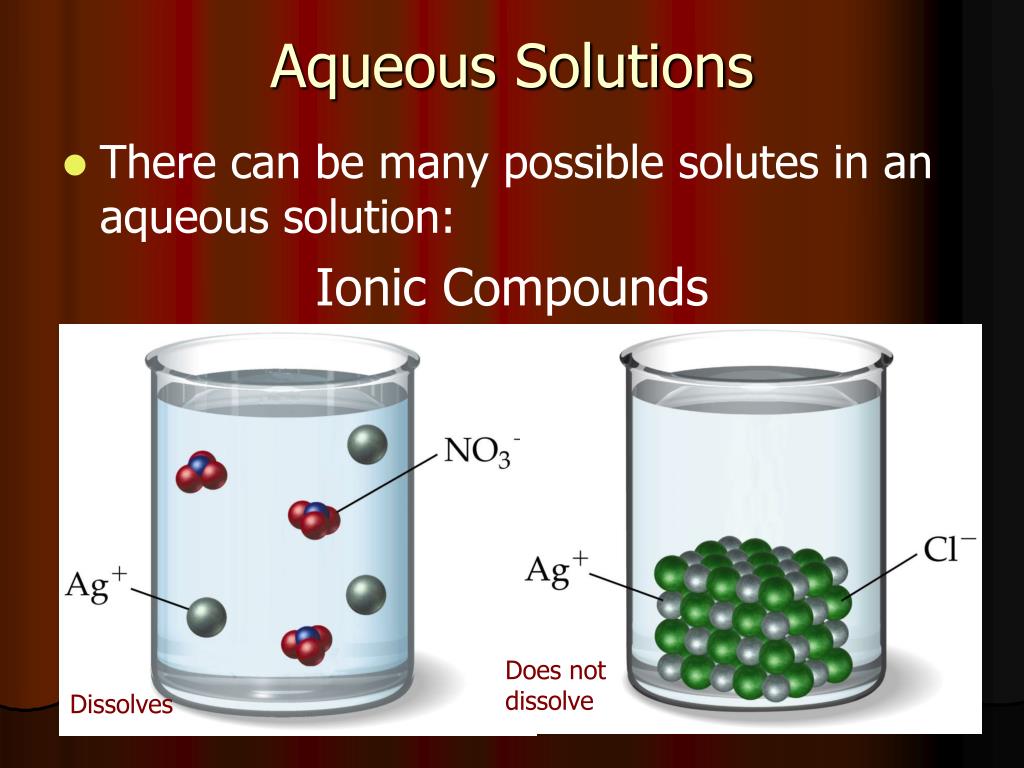
In An Aqueous Solution Do Acids Or Alkalis Accepts Protons

Hey there, science adventurers! Ever wondered what happens when you mix some bubbly stuff with water? It’s like a tiny, invisible party in your glass, and today we’re going to crash it to uncover a super cool secret about two of the main guests: acids and alkalis!
Imagine you’ve got a glass of water, right? It’s pretty chill, just hanging out. Now, let’s sprinkle in a little something special. This is where our story gets exciting, like adding a dash of magic potion to your ordinary lemonade!
We’re going to zoom in on the microscopic world, where tiny, energetic particles are always on the move. Think of them as super-fast dancers at a cosmic rave. And in this rave, there are some very specific dance moves, especially when it comes to something called a proton. Protons are like the party favors, the tiny little bits of attention everyone wants!
Now, our two main characters, acids and alkalis, have very different personalities when it comes to these proton party favors. It’s like deciding who gets the last slice of pizza at a party – everyone has an opinion!
Let’s start with our flamboyant friends, the acids. These guys are like the generous hosts of the proton party. They’ve got protons to spare, and they’re just itching to hand them out!
Think about your morning orange juice. That zesty tang? That’s your acid, getting ready to share its proton goodness. It’s so eager, it’s practically shouting, "Here, have a proton! Take another! Don't be shy!"
When an acid dives into an aqueous solution – that’s just a fancy word for water – it’s like dropping a confetti cannon. Out fly those protons, ready to join the water molecules in their dance. They’re basically saying, "Peace out, proton! Go make some new friends!"

So, in a nutshell, when you add an acid to water, it’s all about giving away protons. They are the proton-donating champions of the chemical world. They’re the ones who start the proton chain reaction!
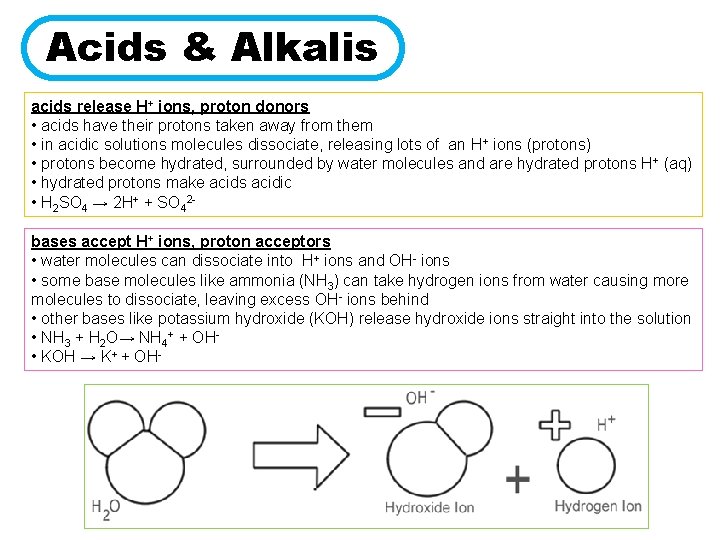
Now, let's switch gears and meet the other half of our dynamic duo: the alkalis. These guys have a completely different vibe. While acids are all about handing out, alkalis are all about collecting!
Imagine your laundry detergent. That smooth, slightly slippery feel? That’s often an alkali at work. And this alkali is like a super-enthusiastic proton magnet!
When an alkali hits the water party, it’s not looking to give anything away. Oh no, it’s looking to grab. It sees those free-floating protons from the acids (or even from the water itself) and thinks, "Ooh, shiny! I’ll take that!"
So, an alkali is like the ultimate proton-hoarder. It’s actively seeking out and accepting protons. It’s the one saying, "Got a proton? Gimme! I've got plenty of space for more!"
This process of accepting protons is a huge deal in chemistry. It’s what defines whether something is an acid or an alkali. It’s their fundamental chemical handshake!
Think of it like a game of hot potato, but with protons. The acid is the one with the proton, and it quickly passes it off. The alkali is the one eagerly waiting to catch it and hold onto it for dear life.
So, who's the proton catcher?
Drumroll, please… It’s the alkalis! They are the ones who are built to grab onto those precious protons.
+solution.+Hydrogen+ion+%3D+lone+proton+H%2B.jpg)
When you have an acidic solution, it’s overflowing with protons that the acid has released. When you add an alkali to that, it’s like throwing a sponge into a proton-filled swimming pool. The alkali just soaks them up!
This is why when you mix a strong acid and a strong alkali, they can neutralize each other. The acid is trying to give away protons, and the alkali is trying to snatch them up. They basically cancel each other out in a very satisfying chemical showdown!
It's a beautiful dance of giving and taking, of donating and accepting. This proton exchange is at the heart of so many reactions, from making your tummy feel better when you have heartburn (thanks, antacids!) to how our bodies function every single second.
So, the next time you’re looking at a bottle of something that fizzes or feels a bit slippery, remember the tiny proton party happening inside. Remember the acids, the generous proton-givers, and the alkalis, the eager proton-receivers.
It’s a simple concept, but it’s incredibly powerful. It’s the microscopic choreography that makes the chemical world go ‘round. And isn’t that just wonderfully fun to think about?
So, to recap for our amazing science fans: Acids donate protons, like they’re handing out flyers at a concert. Alkalis accept protons, like they’re collecting autographs from their favorite pop stars!
This is the core of the Brønsted-Lowry theory, if you want to sound super smart at your next gathering. But don’t worry, the core idea is simple: acids give, alkalis take!
It’s like a chemical high-five, or perhaps more accurately, a chemical proton-pass! The acid initiates the transfer, and the alkali seals the deal by accepting. It’s a perfect partnership, albeit one with a very specific task.
So there you have it! In the splashy, bubbly world of aqueous solutions, it's the alkalis that are the true proton acceptors. They are the rockstars of proton acquisition, always ready to catch that tiny, crucial particle. Isn't science just the coolest?
