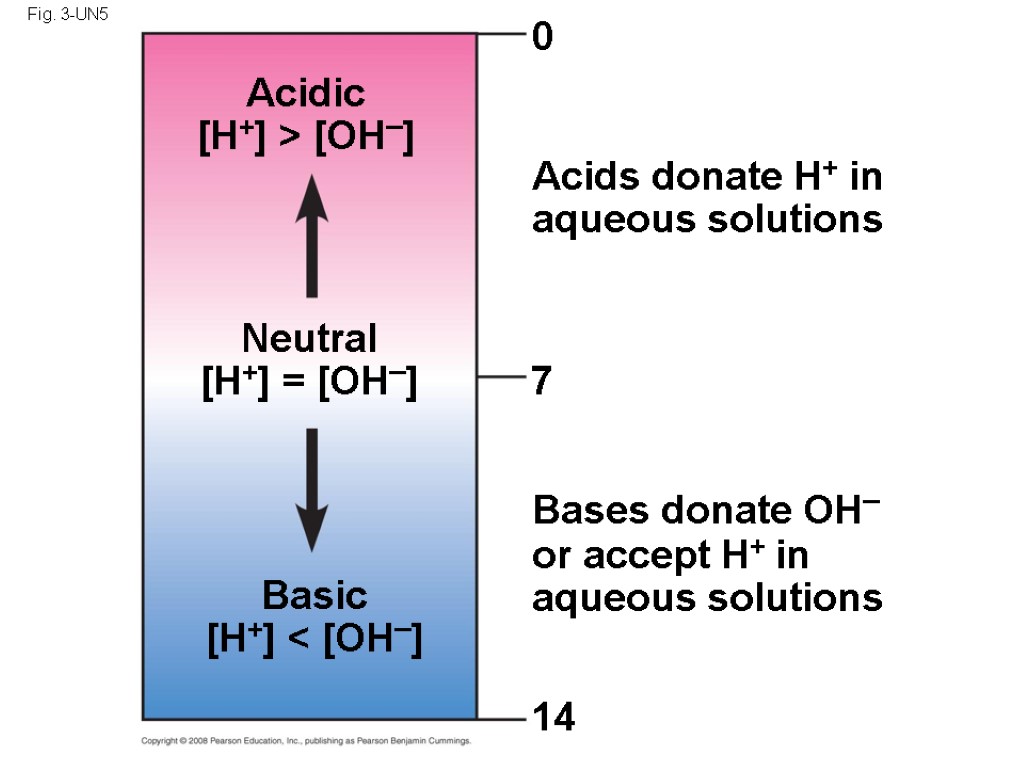
In An Aqueous Solution What Particle Do Acids Donate
Imagine your favorite fizzy drink, the one that tickles your nose and makes your taste buds sing! What’s making that delightful zing? It’s all thanks to tiny, invisible things called acids, and they’re not as scary as they sound. They’re actually quite generous, always looking to share something special when they jump into water.
Think of water as a super friendly, bustling party. When an acid decides to join the fun, it doesn't just plop in and sit there. It brings a gift, a little something it’s eager to give away.
This special gift isn’t a physical object like a toy or a cookie. It’s much smaller, almost like a tiny energy packet. This energetic little guy is called a proton. Sounds fancy, right? But really, it’s just a teeny-tiny bit of electrical charge, like a miniature spark.
So, in the big, watery party, acids are the friendly guests who arrive with a spare proton to hand out. They’re like the ultimate sharers of the microscopic world. They can't wait to find a water molecule that’s ready to accept their donation.
When an acid gives away its proton, it’s like it’s saying, “Here, have this! This is what I do!” It’s this act of donating that makes something an acid in the first place. Without this generosity, it would just be something else entirely.
Think about lemon juice. It’s super sour, right? That sourness is partly because of the citric acid in it. When citric acid meets water, it starts handing out those protons like confetti at a parade.
And vinegar? That sharp, tangy taste? That’s acetic acid at work, also happily donating its protons to the water party. It’s the same story for the acids in your stomach, helping you digest your food.
It’s not just about sour tastes, though. This proton donation is happening all around us, in things we love. That satisfying fizz in your soda? That’s carbon dioxide reacting with water to form carbonic acid, which then, you guessed it, donates its protons!

So, the next time you enjoy a tart berry or a tangy pickle, you can think of the acids inside them performing their generous act. They are literally sharing their protons to make things happen.
It's a bit like a secret handshake between the acid and the water. The acid offers its proton, and the water molecule, often changed slightly by accepting it, is happy to take it.
This might seem very technical, but the core idea is simple generosity. Acids are programmed to share. They can't help but give away this little positive spark when they get wet.
And the water? It’s the perfect host, always ready to receive and accommodate. It's so good at this that it often breaks apart itself to make room for the donated proton.
This proton is actually called a hydrogen ion when it's floating around on its own in water, looking for a friend. It's like a little positively charged wanderer, and it’s the reason why acidic solutions behave the way they do.

So, when we say "acids donate protons," it's really about this fundamental sharing behavior. It's the defining characteristic that makes an acid an acid.
Even in the complex world of chemistry, there are these simple, beautiful stories of interaction and generosity. Acids are the givers, and water is the gracious receiver, creating the chemistry we experience every day.
Think of your favorite tangy yogurt. The lactic acid inside is doing its part, donating protons and contributing to that yummy, slightly tart flavor we enjoy.
This act of donating is so crucial that scientists have different ways of talking about it, but the heart of the matter remains the same: acids give away protons.
It's a concept that, once you grasp it, makes you look at everyday things differently. That splash of lime in your guacamole? Pure proton-donating magic!
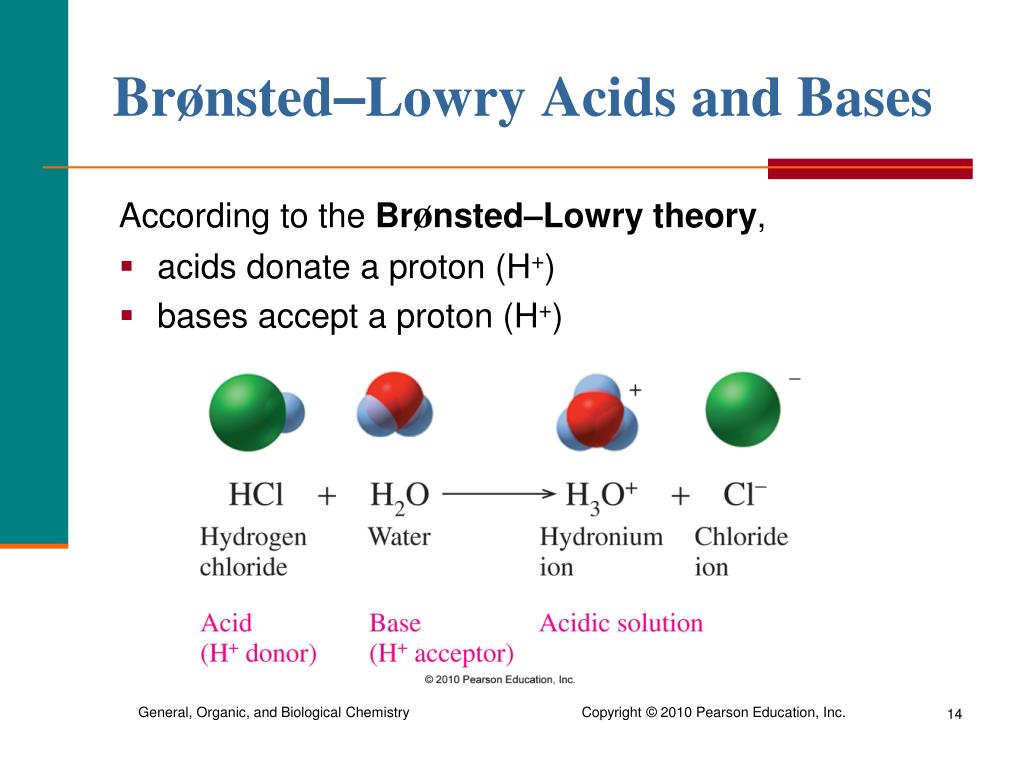
Even the acids that help clean your house are just doing their job by sharing their protons, often to break down dirt and grime. They are the microscopic janitors of the world!
The surprising thing is how these tiny, almost invisible acts create such big, noticeable effects. That tingle on your tongue, that cleaning power, it all starts with a simple proton donation.
So, while we might not see these protons with our own eyes, their impact is everywhere. They are the silent engines behind so many of the flavors and functions we appreciate.
It's like a microscopic act of kindness. The acid, feeling comfortable in its watery environment, finds a purpose in sharing its proton.
And the water molecules, they are not just passive bystanders. They are active participants, ready to embrace the proton and transform.
This makes the whole process a dynamic dance of giving and receiving. It’s a constant exchange happening in every glass of lemonade, every bite of sour candy.
The next time you feel a sharp, pleasant sensation from something acidic, remember the proton. It's the tiny, energetic gift that the acid is so happy to share.
It’s a reminder that even the most complex science can be boiled down to simple, heartwarming acts. Acids are the generous friends of the aqueous world, always ready to donate their protons.
So, cheers to the acids and their protons! They bring the zing, the tang, and a whole lot of fun to our world, one tiny donation at a time.
It’s a beautiful illustration of how fundamental actions at the smallest scale can lead to the experiences we cherish the most.
And that, in a nutshell, is what acids are so good at in water: sharing their protons with joy!
