Ionic Lattices Are Structures Formed By Ionic Compounds.

Ever wonder what holds everyday things together? Like, really, really together? Think about a salt shaker, that humble dispenser of flavor. What's going on inside those tiny white crystals? It's not just a pile of salt! It's an amazing, organized structure called an ionic lattice. And guess what? It's way cooler than it sounds.
Imagine a party. A super organized, never-ending party. That's kind of like an ionic lattice. It's a structure built by ionic compounds. So, what's an ionic compound? Well, it's a team-up of two different types of atoms, usually a metal and a nonmetal. They're not just hanging out; they're bonded together in a very special way. Think of it like giving and taking. One atom basically gives away one or more of its tiny parts, called electrons, to another atom. This makes them become charged little buddies.
Now, here's where the magic happens. When atoms become charged, they get these cool tags: one becomes positively charged (we call it a cation) and the other becomes negatively charged (a anion). And what do opposite charges do? Yep, they attract each other! It's like little magnets. This strong attraction is what holds them together.
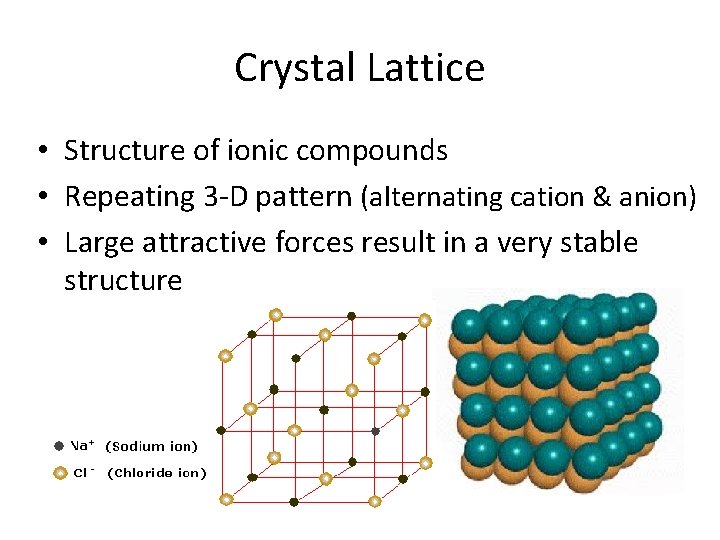
But it's not just two atoms stuck together. Oh no, that would be too simple. In an ionic compound, these charged atoms arrange themselves in a super neat, repeating pattern. It’s like building with LEGOs, but instead of colorful plastic bricks, you have these positively and negatively charged ions, all lining up perfectly. The positive ions love being surrounded by negative ions, and the negative ions love being surrounded by positive ions. It's a big, beautiful, three-dimensional arrangement where everyone gets along (in a charged sort of way!).
This repeating, geometric structure is the ionic lattice. It’s the fundamental blueprint for many solid substances we use every single day. Salt, or sodium chloride (NaCl), is the classic example. You've got positive sodium ions (Na+) and negative chloride ions (Cl-) all snuggled up together in this organized grid. It’s not just a random jumble; it’s an architecture of attraction!
Why is this so entertaining? Because it's hidden! You can't see this incredible organization with your naked eye. It's happening at a microscopic level, shaping the properties of everything from your sugar to your garden soil. Think of it as a secret society of atoms, working together to create the solid world around us. It's like finding out that your favorite cookie isn't just flour and sugar, but a tiny city of ions holding it all together!
What makes it special? The incredible strength and order. This arrangement is super strong. It takes a lot of energy to break apart an ionic lattice. That's why things like salt and sugar are solids at room temperature and don't just melt away when you look at them. They’re built to last, thanks to this powerful, orderly structure.
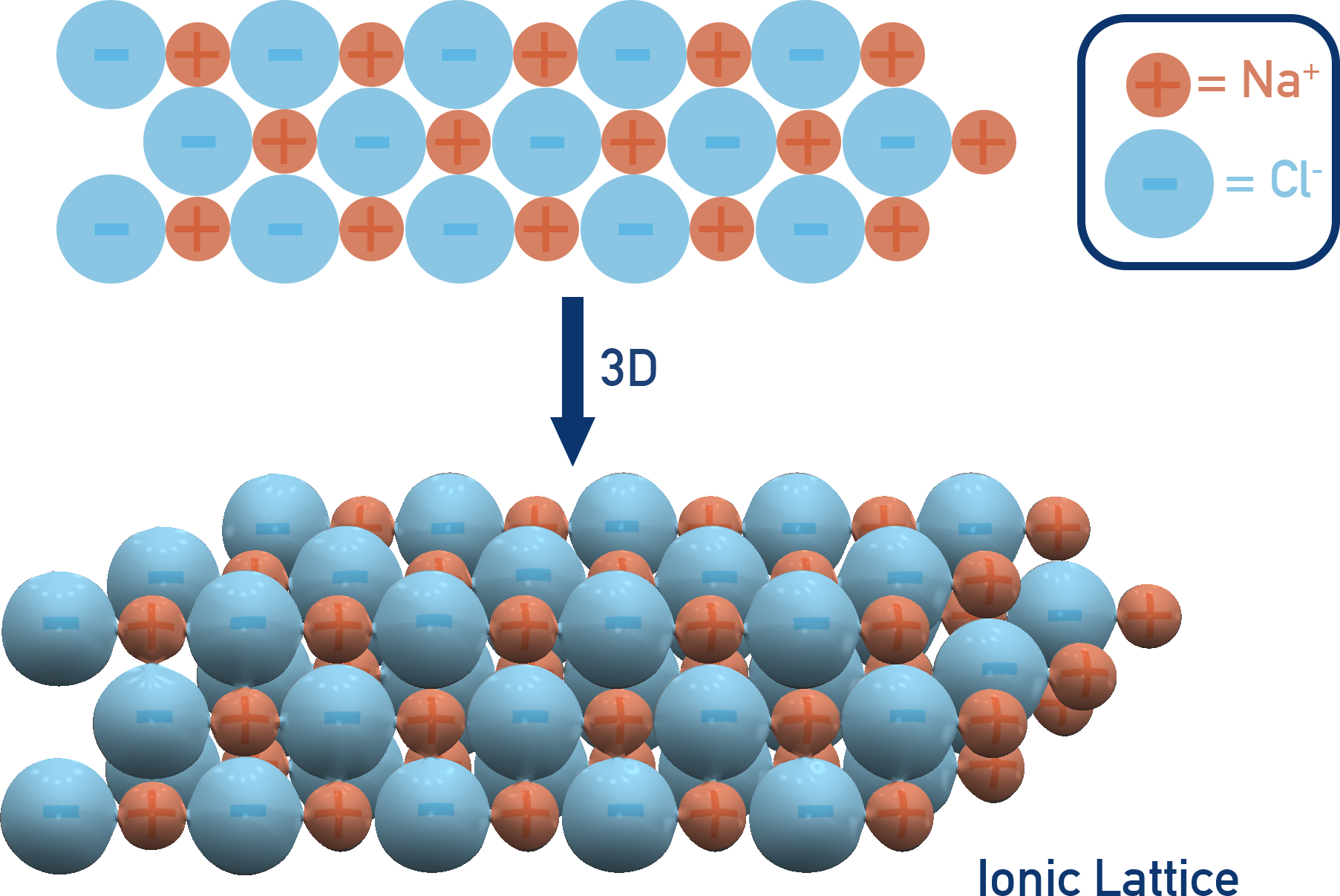
Imagine tiny, perfectly stacked cubes, but each cube is made of alternating positive and negative charges. That's a simplified view of a cubic ionic lattice, like the one in table salt. But it’s not always cubes! Depending on the ions involved and their sizes, these lattices can take on all sorts of fascinating shapes and arrangements. It’s like nature’s own crystal builders, meticulously crafting these structures.
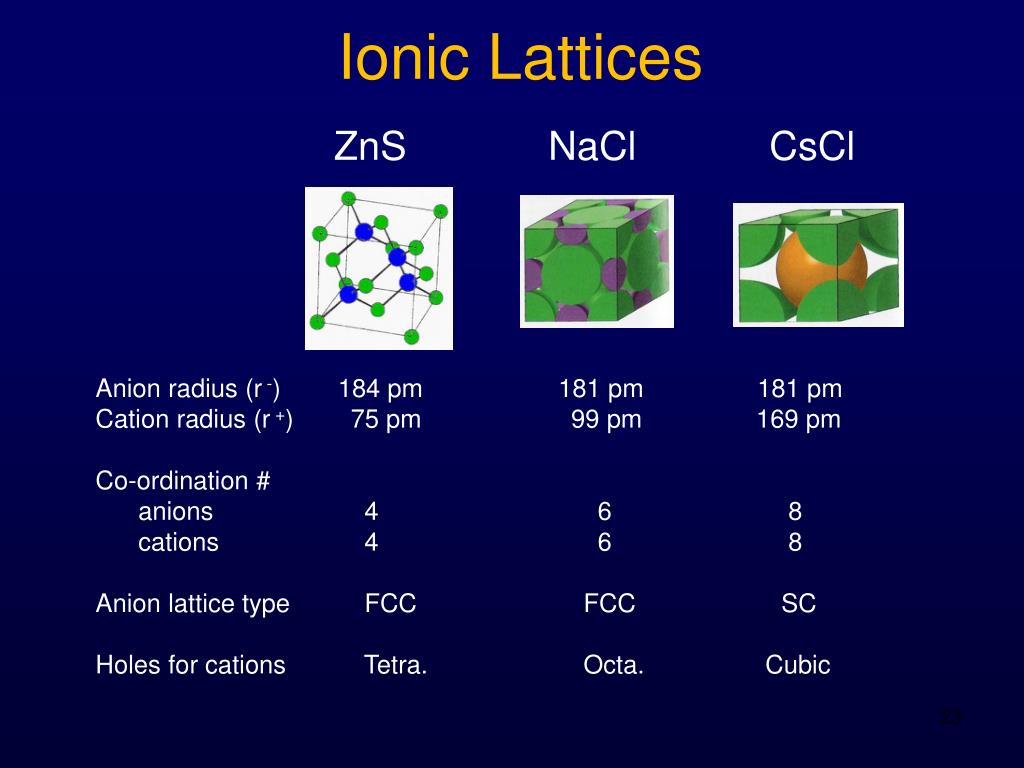
The beauty of the ionic lattice is its simplicity and its universality. While the specific ions and their arrangement can differ, the underlying principle of electrostatic attraction and the formation of a repeating, three-dimensional structure is the same. It's a fundamental concept in chemistry that explains so much about the materials we interact with.
Think about how a tiny pinch of salt can enhance your food. That flavor comes from the sodium and chloride ions. But before they deliver that taste, they've been through this incredible journey of becoming charged and forming this organized lattice. It’s like they’ve been training for their flavor-delivering mission!
The forces holding these lattices together are called ionic bonds. These aren't like the bonds in a friendly handshake; these are powerful attractions that dictate how these compounds behave. They are responsible for their high melting points, their ability to dissolve in water (because water molecules can gently pull these ions apart), and their hardness.

It's truly amazing to think that the seemingly simple, solid substances around us are the result of such a sophisticated and beautiful microscopic architecture. The next time you pick up a piece of chalk, a sugar cube, or even a piece of glass, remember the unseen world of ionic lattices, the unsung heroes of solid materials, quietly holding everything together in a perfectly organized dance of charges.
It's a fascinating world of atomic architects, building structures that are both strong and elegant. Ionic lattices are the hidden blueprints for so much of our material reality!
So, why should you be curious? Because this knowledge unlocks a new way of seeing the world. It turns everyday objects into fascinating scientific marvels. You start to appreciate the intricate order that underlies the apparent simplicity. It's like discovering a secret code that explains how things are made. And who doesn't love a good secret code?
If you're intrigued, dive a little deeper! Look up some pictures of different ionic lattice structures. You'll see beautiful, geometric patterns that are truly a feast for the eyes, even if you need a microscope to see them. You might find yourself looking at a simple salt crystal with a whole new sense of wonder. It’s a journey from the mundane to the magnificent, all thanks to the power of ionic attraction and the beauty of organized structures!
