Is Enthalpy Change Of Solution Exothermic Or Endothermic
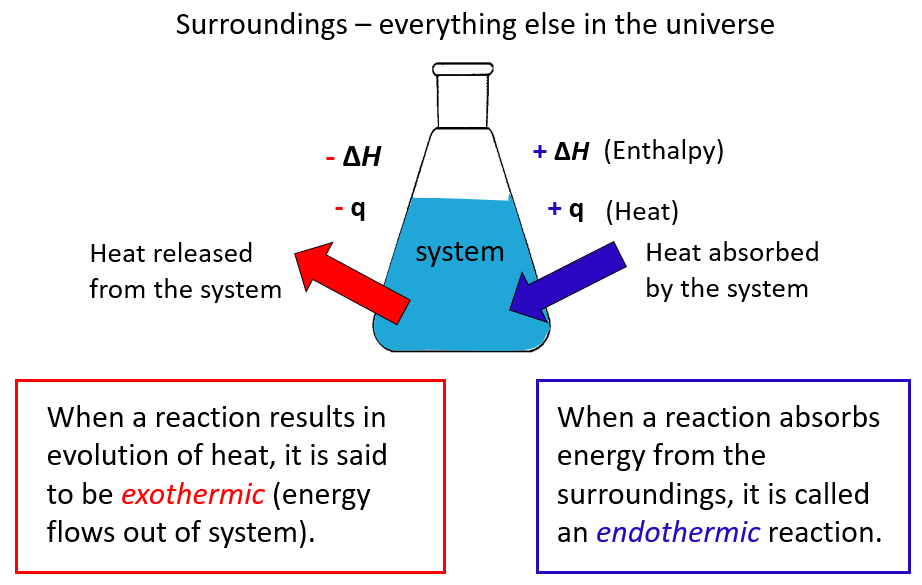
Ever found yourself staring into a glass of water, a little packet of something in your hand, and wondered what magical science is about to happen? You know, when you mix stuff together and things get a bit… lively? We're talking about the amazing world of enthalpy change of solution, and it’s way more exciting than it sounds, I promise!
Imagine you're having a super chill party, and you decide to make some refreshing lemonade. You grab your trusty pitcher, some water, and a generous scoop of sugar. You stir, and stir, and stir. What usually happens? The sugar disappears like a ninja in the night, becoming one with the water. Pretty neat, right? This process of dissolving things is what scientists lovingly (and perhaps a little dramatically) call the enthalpy change of solution. Think of it as the “mood swing” of the dissolving process. Will it be a happy, energetic mood or a quiet, sleepy one?
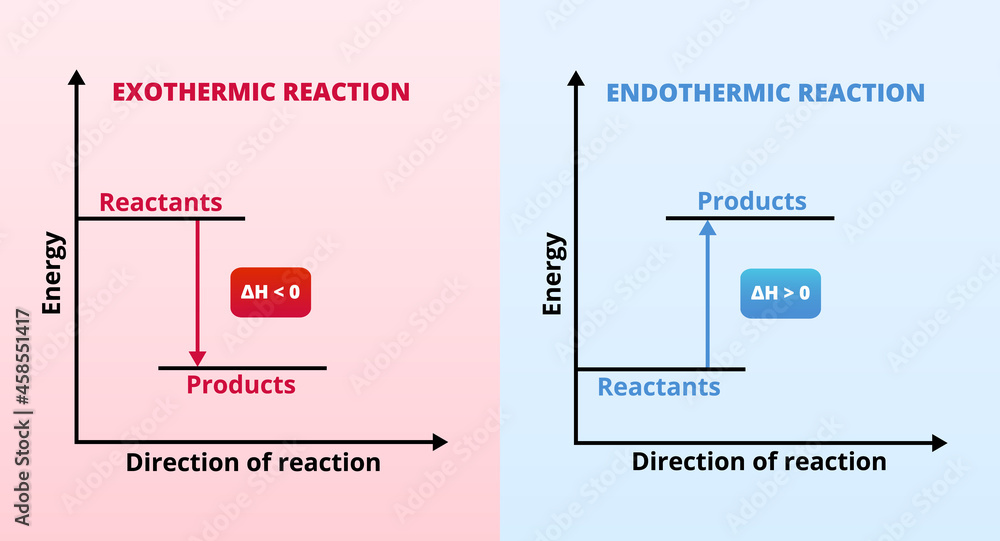
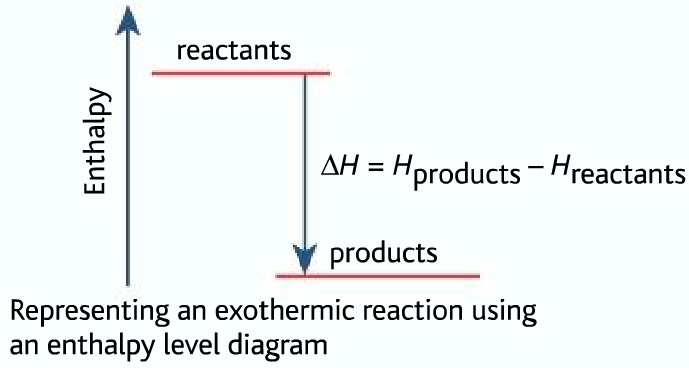
Now, sometimes, when you dissolve something, the solution gets wonderfully, gloriously warm. It’s like your drink is giving you a little hug! This is when the enthalpy change of solution is exothermic. “Exothermic” sounds like a fancy word, but it just means “giving off heat.” So, when you dissolve something and the mixture feels warm to the touch, it’s like the molecules are doing a little happy dance and throwing off some extra energy as heat. Picture this: you’re in the middle of a blizzard, and you dissolve some special magic crystals in your water bottle. Suddenly, your hands are toasty warm! That’s exothermic power at work, folks!
Our friend, sodium hydroxide, is a superstar when it comes to being exothermic. If you’ve ever seen one of those hand warmers you can buy in a pinch, they often rely on a similar principle. Dissolve a bit of this stuff, and BAM! Instant warmth. It’s like the molecules are saying, "Here, have some cozy vibes!" So, if you’re ever making a hot beverage and decide to add something that makes it feel like it just came off the stove, you’re witnessing an exothermic enthalpy change of solution. It’s nature’s way of saying, "Here's a little extra sunshine for your drink!"
But then, there’s the other side of the coin. Sometimes, when you dissolve something, the solution gets… well, a little bit chilly. It’s like your drink decided it needs some personal space. This is when the enthalpy change of solution is endothermic. “Endothermic” is the opposite of exothermic – it means the process absorbs heat from its surroundings. So, if the solution gets cold, it’s because the dissolving molecules are so busy breaking apart and getting cozy with the solvent that they’re actually sucking up all the heat from the mixture. It's like they're so engrossed in their newfound friendship that they forget to keep things warm!
Think about when you use an instant cold pack. You know, the kind you crack and then it gets ice cold? That’s a classic endothermic reaction at play! You’re dissolving something that’s so eager to find new friends (the solvent molecules) that it’s snatching heat away from everything around it, including your own skin (don't leave it on too long, though!).
A common culprit for endothermic behavior is ammonium nitrate, the stuff often found in those instant cold packs. When it dissolves in water, it’s so enthusiastic about its new surroundings that it practically inhales the heat. The result? A delightfully (or perhaps alarmingly) cold concoction. Imagine you’re feeling a bit feverish, and you dissolve some special powder in your water. Instead of a warm hug, you get a refreshing, cool wave. That’s endothermic magic!
So, to sum it up in the most non-science-y way possible: when you dissolve something, it can either make your drink feel like a warm hug (exothermic) or a refreshing ice pack (endothermic). It all depends on the personalities of the dissolving particles and the solvent molecules. Are they feeling generous and giving off heat, or are they a bit needy and soaking it all up?
It's a bit like a party. Sometimes the music is so good, everyone starts dancing and getting all hyped up, radiating energy – that’s exothermic! Other times, everyone’s so engrossed in deep conversations and sharing secrets that they’re a bit of a quiet, absorbing bunch – that’s endothermic! The same goes for dissolving. It's all about energy exchange. You're not just mixing ingredients; you're witnessing a tiny, energetic dance party happening right in your glass. Pretty cool, huh? So next time you're dissolving something, pay attention to how it feels. Is it giving you a warm fuzzy, or a cool surprise? You’re basically a scientist at home, and that’s something to feel good about!
