Is Rubbing Alcohol The Same As Isopropyl Alcohol

You know those moments when you’re elbow-deep in a DIY project, or maybe just trying to clean up a sticky spill that’s defying all logic, and you reach for that trusty bottle of “rubbing alcohol” from the medicine cabinet? Yeah, me too. For the longest time, I just assumed it was all one glorious, germ-busting entity. But then, something sparked my curiosity. I was at the store, staring at a shelf packed with bottles, and I saw “rubbing alcohol” and right next to it, “isopropyl alcohol.” My brain did a little wobble. Wait a sec, are these two different things? Or are they just… friends?
It’s kind of like when you’re trying to figure out if your cousin twice removed is actually your second cousin. The terms get tossed around, and you end up just nodding along, hoping no one asks you to draw a family tree. So, I decided it was time to get to the bottom of this whole alcohol situation. Because honestly, if I’m going to be disinfecting my phone or trying to get permanent marker off my kitchen counter (don't ask), I want to know exactly what I'm dealing with, right?
The Great Alcohol Identity Crisis
Let’s dive right in. The short, sweet, and slightly anticlimactic answer is: mostly, yes. In the vast majority of everyday situations, when you grab that bottle labeled “rubbing alcohol,” you are, in fact, grabbing a bottle of isopropyl alcohol. It’s like calling your best friend by their nickname. Everyone knows who you mean, even if their actual name is something a bit more formal.
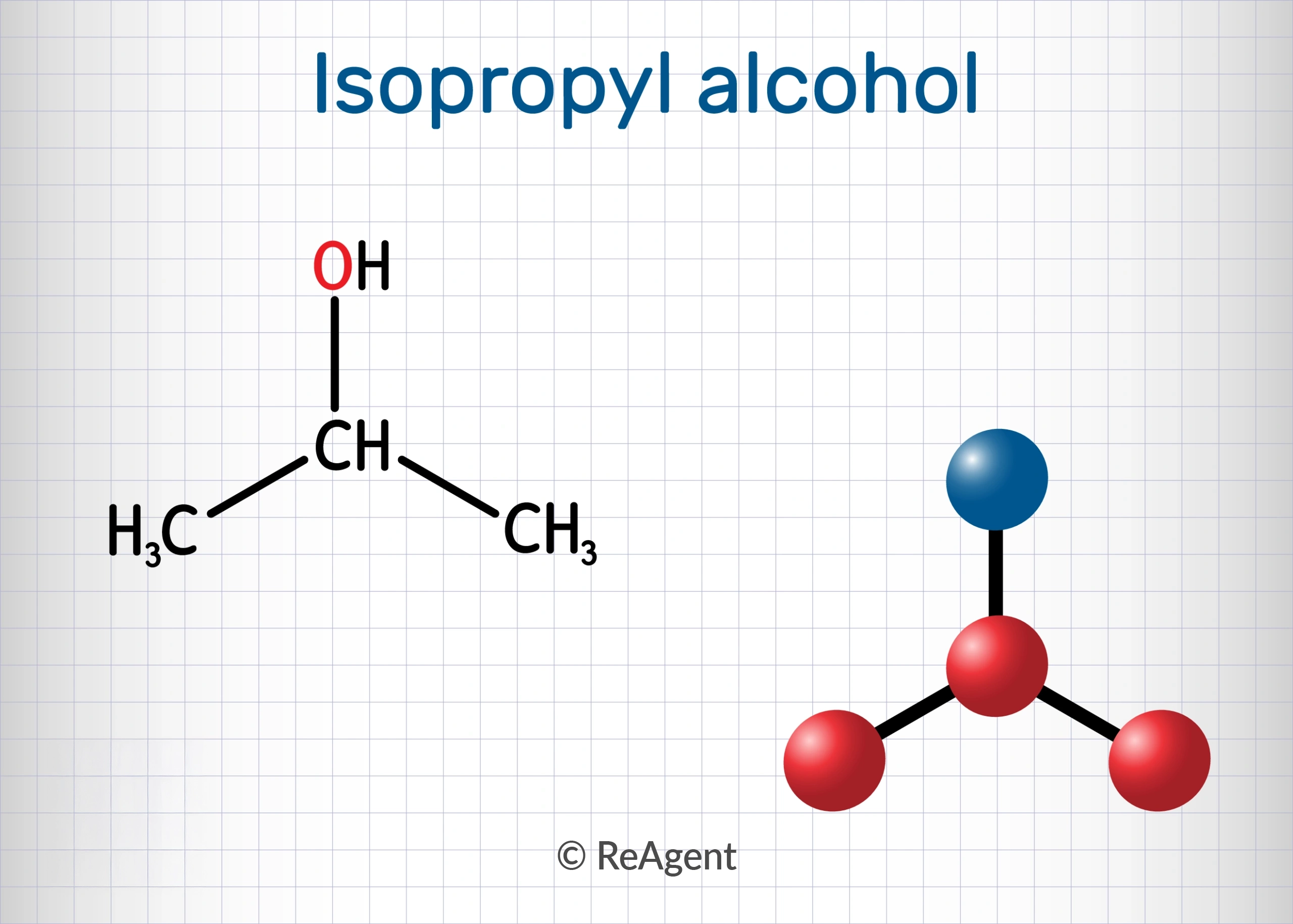
Think of it this way: Isopropyl alcohol is the ingredient, and rubbing alcohol is the product name. It's the brand name vs. the generic drug, but with a chemical twist. Most rubbing alcohol you find in drugstores, supermarkets, and even dollar stores is formulated with a high percentage of isopropyl alcohol, typically around 70% or 91%.
Why is it so prevalent? Well, isopropyl alcohol is a fantastic disinfectant and solvent. It’s relatively inexpensive to produce, readily available, and it does a darn good job at what it’s supposed to do: kill germs and break down sticky stuff. So, manufacturers slapped “rubbing alcohol” on the label because, well, that’s what people use it for! It’s descriptive, it’s catchy, and it gets the job done.
So, What About That "Rubbing" Part?
This is where it gets a tad interesting. The term “rubbing alcohol” itself implies its intended use – for rubbing on the skin. This is important because not all alcohols are created equal when it comes to safety for topical application. Some alcohols, like denatured alcohol (which we’ll get to in a sec!), are absolutely not meant for your skin. They can be quite toxic.
Isopropyl alcohol, however, has a long history of use as an antiseptic. It’s effective at killing bacteria, fungi, and viruses, making it a staple in first-aid kits and healthcare settings for cleaning wounds (though now usually diluted and after the initial injury) and sterilizing equipment. When it's formulated as rubbing alcohol, it’s typically diluted with water to create a safe and effective concentration for skin application.
The 70% solution is often preferred for disinfection because it’s more effective at killing microbes than higher concentrations. Why? It's a bit of a chemical dance. The water in the 70% solution helps the alcohol penetrate the cell walls of bacteria, while the alcohol itself denatures the proteins within those cells, effectively killing them. A higher concentration of pure alcohol might evaporate too quickly before it can do its job properly, or it might not penetrate the cell as effectively.
So, the next time you see that bottle of 70% isopropyl alcohol, you can confidently think, "Ah, that's the good stuff for getting rid of those pesky germs!" It's not just a random chemical; it's a targeted germ-fighting tool.
Where the Lines Get Blurry (But Not Too Blurry)
Now, let’s acknowledge the slight nuance. While isopropyl alcohol is the star of the rubbing alcohol show, are there ever other things in that bottle? Generally, no, not in the stuff you buy for household use. However, some historical or industrial formulations of “rubbing alcohol” might have included other alcohols.
The most common other alcohol you might encounter, and one that people sometimes confuse, is ethanol. Ethanol is the type of alcohol found in alcoholic beverages (like beer, wine, and spirits). And guess what? Ethanol is also a pretty decent disinfectant!

You might see bottles labeled “ethanol rubbing alcohol” or even just “alcohol” for industrial cleaning purposes. The key difference is that ethanol is what we drink (in carefully controlled amounts, of course!), while isopropyl alcohol is strictly for external use. You absolutely, positively, never want to ingest isopropyl alcohol. It can be toxic and cause serious health problems. So, let this be your friendly reminder: keep it on the outside!
In some regions or for specific purposes, you might also find “denatured alcohol.” This is essentially ethanol that has additives to make it unfit for human consumption. This is often used as a solvent or cleaner in industrial settings, and it is definitely NOT for rubbing on your skin. So, if you see “denatured alcohol,” treat it with caution and keep it far away from your body.
But back to our friendly neighborhood rubbing alcohol. For the most part, the bottles you’re picking up are going to be primarily isopropyl alcohol, with water as the diluent and maybe a tiny bit of something to keep it from evaporating too fast or to make it slightly less harsh on the skin (though that’s rare in the basic stuff).
It’s like the difference between a cola and a generic cola. Both are fizzy, brown, and often satisfy the craving, but one might have a secret ingredient or a slightly different flavor profile. Isopropyl alcohol is the universally recognized champion for rubbing alcohol purposes.

Why the Different Concentrations?
You’ve probably noticed that rubbing alcohol comes in different strengths. The most common are 70% and 91% (or sometimes 99%). As we touched on earlier, the 70% solution is generally considered the gold standard for killing germs due to the water content facilitating better penetration and action. So, why the 91% or 99%? What’s the point?
Well, higher concentrations of isopropyl alcohol are fantastic for different jobs. They are more potent solvents. Think about trying to remove stubborn sticky residue from labels, or cleaning electronics where you want the alcohol to evaporate very quickly without leaving behind any moisture. For those tasks, a higher concentration is your best bet. It evaporates almost instantly and is less likely to damage delicate components.
It's kind of like having a toolkit. You wouldn’t use a hammer to tighten a screw, right? Similarly, you wouldn’t use 70% rubbing alcohol to quickly clean a dusty circuit board. The higher concentration evaporates faster, leaving less residue and reducing the risk of electrical shorts. So, while 70% is your germ-killing buddy, 91% or 99% is your super-powered cleaning and solvent sidekick.
It’s also worth noting that the higher concentrations can be more irritating to the skin, so if your goal is simply to disinfect a minor cut (after cleaning it properly with soap and water, of course!), the 70% is generally recommended. It’s all about choosing the right tool for the job!

The Bottom Line: Your Trusted Friend
So, to wrap it all up in a neat little bow, is rubbing alcohol the same as isopropyl alcohol? Yes, for all practical purposes when you're buying it for home use. Rubbing alcohol is essentially a product name for a solution of isopropyl alcohol and water, typically at a concentration of 70% or 91%.
It’s the go-to for cleaning minor wounds (after initial cleaning!), disinfecting surfaces, and tackling all sorts of household messes. It’s a workhorse of the cleaning and first-aid world.
The confusion often arises because the term "alcohol" is broad and can encompass many different chemical compounds. But in the context of that familiar bottle in your bathroom cabinet, you can rest easy knowing you’re dealing with good old isopropyl alcohol.
So, next time you reach for it, you can do so with a little more knowledge and a lot less confusion. You're not just grabbing a bottle of "rubbing alcohol"; you're wielding the power of isopropyl alcohol, a trusted ally in the fight against germs and grime. And isn't that kind of empowering? Now go forth and disinfect (responsibly, of course)!
