Methanol Density Mg Ml
Okay, let's talk about something super exciting. No, seriously! We're diving headfirst into the world of Methanol Density. And before you start yawning and picturing yourself back in a very dull science class, hold on a sec. This is going to be fun. I promise. Think of it as a tiny adventure for your brain. We're talking about Mg/mL, which sounds fancy, but it’s just a way to measure how much "stuff" is packed into a tiny space. Like how many marshmallows can you squish into a little box.
Now, my unpopular opinion? I think Methanol Density is secretly kind of cool. I know, I know. You’re probably thinking, “Who says that?” Well, me! And maybe a few other delightfully nerdy people out there. But seriously, let’s break it down. Imagine you have a little bottle. And you pour some methanol into it. That methanol has a certain weight, right? And it takes up a certain amount of space. The density is just that ratio – how heavy it is for the space it fills.
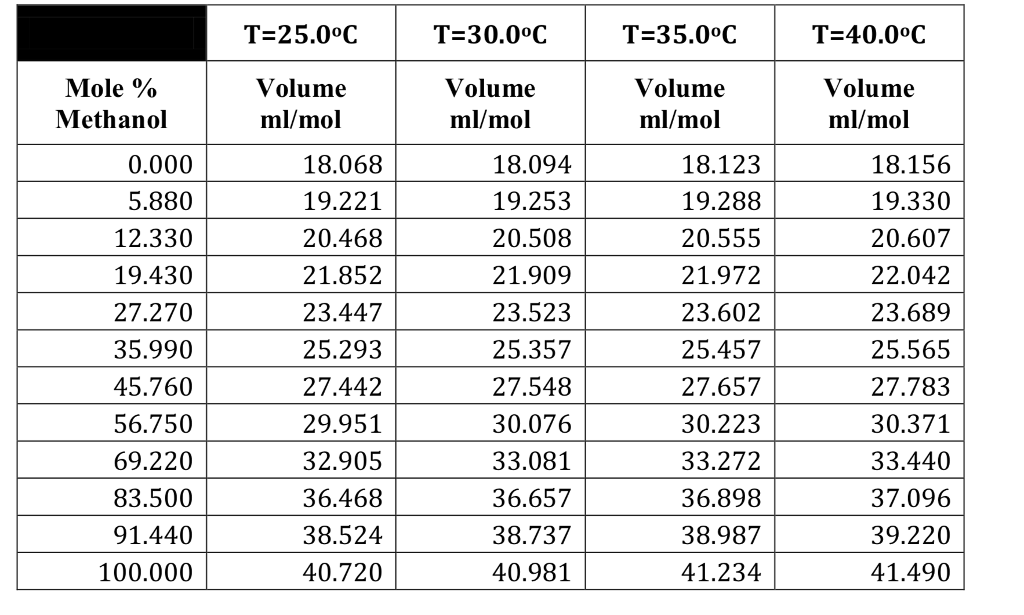
And the number? The specific number for Methanol Density in Mg/mL? It’s somewhere around… let’s say, roughly 0.792 Mg/mL. Yep. That’s the magic number for pure methanol at a comfortable room temperature. What does that mean in plain English? It means methanol is lighter than water. Ever see oil and water separate? Methanol is more like the oil in that scenario. It doesn't quite sink as deeply.
Think about it. Water, our trusty old friend, has a density of about 1 Mg/mL. So, if you had the same size container, you could fit more water in it than methanol. Or, to put it another way, a certain amount of methanol would weigh less than the same amount of water. It’s like comparing a fluffy cloud to a bowling ball. Both take up space, but one is definitely lighter. And methanol, my friends, is a bit more of a cloud.
Why is this even a thing to get excited about? Because these tiny details, these little measurements, are the building blocks of so much. They’re what make things work. When chemists are mixing potions (okay, maybe not potions, but important chemicals!), they need to know how much of each ingredient they’re using. And density is a big part of that puzzle.

Imagine you’re baking. You’ve got flour and sugar. They both take up space in your measuring cup, but they weigh different amounts. If you were trying to make a super precise recipe, you’d need to know those differences. Methanol is no different. Scientists and engineers rely on this Methanol Density Mg/mL figure all the time. They use it to calculate how much methanol they need for a reaction, how it will behave in a mixture, or even how much of it they can safely store.
It’s the quiet hero of many a scientific experiment, the unsung star of the lab bench, the number that makes complex calculations… well, slightly less complex!
Methanol - Density and Specific Weight vs. Temperature and Pressure
And let’s be honest, isn’t there something inherently charming about a liquid that’s a little lighter than water? It’s like it’s got a secret spring in its step. It’s not bogged down by the usual density. It’s just… floating through life with a bit more ease. This is the kind of thinking that gets me through those long afternoons.
So, the next time you hear someone casually mention Methanol Density, give them a knowing nod. You, my friend, are in on the secret. You understand the subtle elegance of 0.792 Mg/mL. You appreciate the fact that a substance can have a distinct weight for its volume, and that this seemingly small piece of information is actually a pretty big deal in the grand scheme of things. It’s not just a number; it’s a characteristic, a personality trait for a chemical.
Think of all the things methanol is used for. Fuel additives, solvents for paints and varnishes, even in some windshield washer fluids to prevent freezing. Behind all those practical applications are these fundamental properties, like its density. It’s the invisible hand guiding its behavior. It’s what helps it mix with other things, or not mix. It’s what dictates how it will flow.
And honestly, the unit itself, Mg/mL, has a certain ring to it, doesn’t it? Milligrams per milliliter. It sounds official, important. It’s like a secret handshake for people who appreciate the finer, more precise aspects of the universe. We're not talking about pounds or ounces here. We're talking about the delicate dance of milligrams and milliliters. It’s sophisticated.
So, here’s to methanol. Here’s to its not-so-surprising density. And here’s to us, the people who find a little bit of fun in understanding how the world around us is put together, one tiny, dense measurement at a time. It’s a small thing, this Methanol Density Mg/mL, but it’s a pretty neat thing if you ask me. And I am, indeed, asking you to agree.

