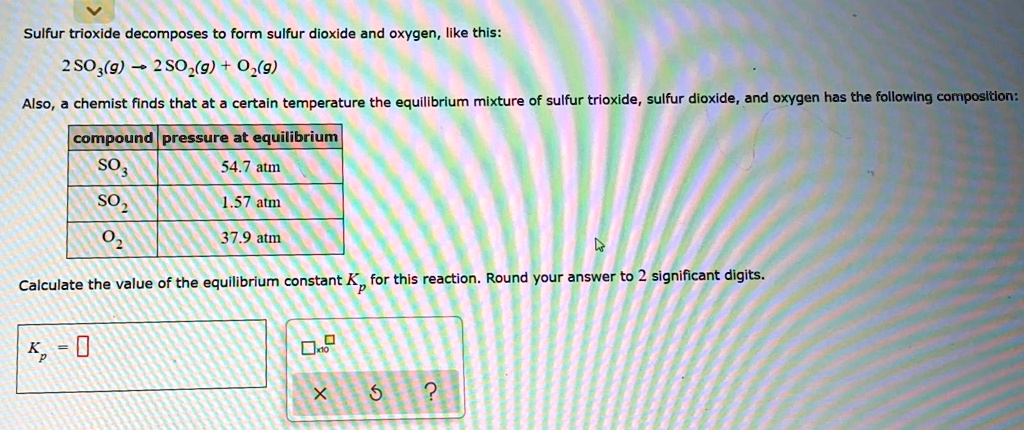
Sulfur Trioxide Decomposes On Heating To Form An Equilibrium Mixture

So, imagine you've got this super energetic molecule, like a party animal that just can't sit still. This particular party animal is called Sulfur Trioxide, or SO₃ for short. It's got a reputation for being a bit… unstable when things heat up. Like a toddler who’s had too much sugar, it just has to break into smaller pieces!
Now, what happens when this energetic SO₃ gets a bit too much of a good thing – meaning, it gets heated up? Well, it decides to have a little party of its own, and in the process, it splits into two other, slightly less frantic molecules. Think of it like a superstar performer who, after a blazing solo, decides to share the stage with a couple of backing dancers. These dancers are Sulfur Dioxide (SO₂) and a tiny little sparkler called Oxygen (O₂).
So, the grand performance looks something like this: SO₃, with a flourish of heat, turns into SO₂ and O₂. Pretty neat, right? It’s like a magician pulling rabbits out of a hat, except these are molecules and the hat is a hot oven!
But here’s where things get really interesting, and honestly, a little bit like a cosmic dance. This isn’t just a one-way ticket to decomposition town. Oh no. These molecules, once formed, start looking at each other and thinking, "You know what? That SO₃ thing looked kind of cool. Maybe we should try putting ourselves back together!"
So, while the SO₃ is busy breaking apart into SO₂ and O₂, the SO₂ and O₂ are simultaneously trying to reform back into SO₃. It’s like a never-ending game of molecular tag! They’re all zipping around, some breaking apart, some coming together, all at the same time, especially when the heat is just right.
This, my friends, is what scientists, in their wonderfully precise way, call an equilibrium mixture. Don’t let the fancy name scare you! Think of it as a really lively, bustling marketplace. In one corner, you’ve got stalls where people are handing out raw materials (that’s our SO₂ and O₂). In another corner, you’ve got stalls where people are assembling those raw materials into something a bit more complex (that’s our SO₃). And the whole place is buzzing with activity, with people moving back and forth constantly. At any given moment, you’ll see a certain number of people buying, and a certain number of people selling. The overall vibe of the marketplace stays pretty consistent, even though individuals are always on the move.
In our sulfur trioxide situation, the “vibe” of the marketplace is the equilibrium. It’s not that the reaction has stopped; it’s just that the rate at which SO₃ is breaking apart is exactly the same as the rate at which SO₂ and O₂ are coming together. It’s a perfect, delicate balance, like a tightrope walker maintaining their composure mid-air. They’re constantly making tiny adjustments, but from our perspective on the ground, they look perfectly still.

So, if you were to peek into this hot, steamy world of sulfur trioxide decomposition, you wouldn't just see SO₂ and O₂ chilling out. You’d see a fantastic mix of SO₃, SO₂, and O₂ all coexisting happily. Some molecules are in their “split” state, others are in their “rejoined” state, and the whole ensemble is in this state of dynamic harmony. It's like a perfectly choreographed dance routine where everyone is moving, but the overall picture remains constant.
It’s not a static pause; it’s a lively, ongoing dance of molecules, all happening at a breakneck speed!
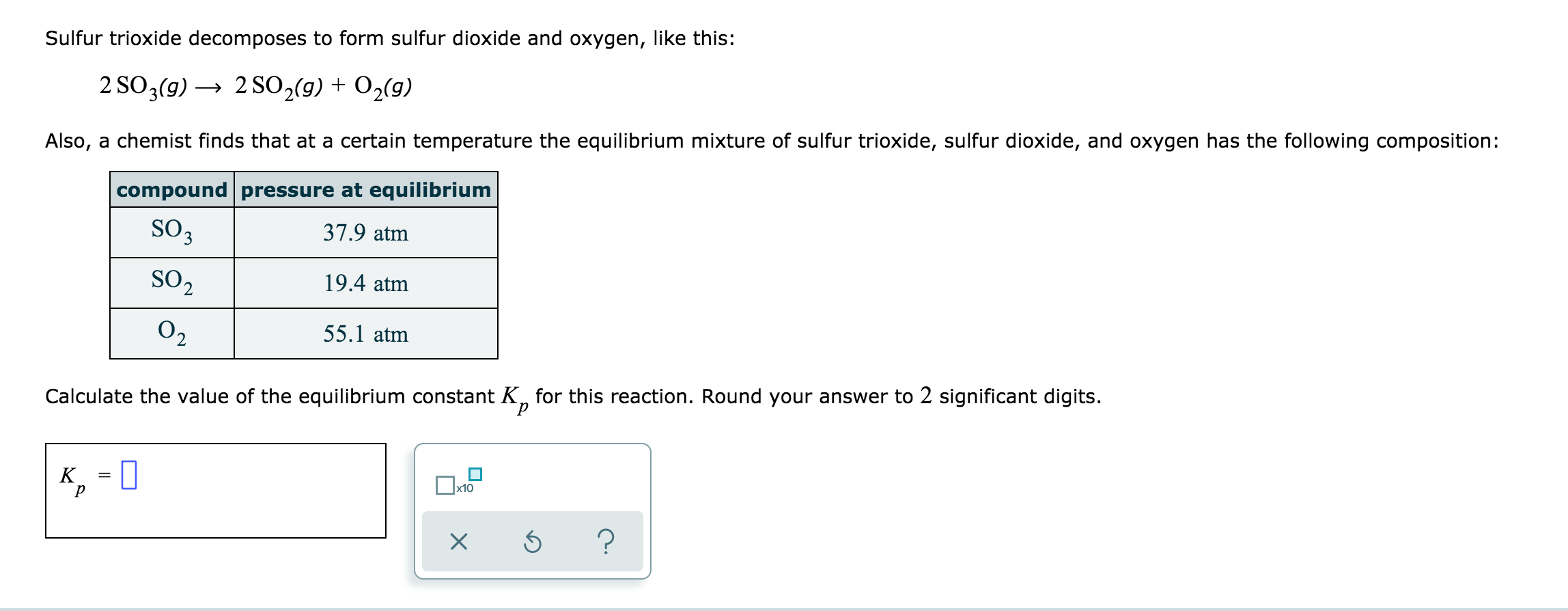
SOLVED: Sulfur trioxide decomposes to form sulfur dioxide and oxygen
And the best part? This balance isn't fixed forever. If you decide to change the conditions – say, you cool things down a bit, or you add more of one of the ingredients – the whole dance floor shifts. The equilibrium might lean more towards breaking apart, or it might lean more towards coming together. It’s like gently nudging one dancer on the stage; the whole group has to adjust to maintain their balance!
So, next time you think about chemical reactions, remember our friend Sulfur Trioxide. It’s not just a boring old chemical formula. It’s a tiny, energetic ball of potential that, when heated, throws a dazzling decomposition party, only to find that its guests are so friendly they decide to form a new party right back! It’s a beautiful illustration of how things in the universe are rarely just one thing or another, but often a vibrant, bustling mix of possibilities, all in a state of perpetual, fascinating equilibrium.