Sulphuric Acid And Sodium Hydroxide Balanced Equation
Ever felt like your kitchen sink was staging a silent protest, refusing to let go of that stubborn grease clog? Or maybe you’ve wondered what makes those little white packets of stuff that come with everything, from new shoes to fancy electronics, so good at soaking up moisture? Well, buckle up, buttercup, because we're about to dive into a world of slightly-less-scary chemistry that’s actually happening all around you, even when you're just trying to make toast. Today’s star players? Sulphuric acid and sodium hydroxide. Don’t let the fancy names fool you; these guys are the unsung heroes (and sometimes villains!) of our everyday lives.
Now, I know what you’re thinking: “Chemistry? Acid? Base? Isn’t that the stuff that eats through metal and makes lab coats smoke?” And yes, in concentrated forms, they absolutely can! Think of the most aggressive cleaning product you’ve ever encountered. It might have a distant cousin in the sulphuric acid family. And sodium hydroxide? Well, it’s the secret ingredient in some seriously powerful drain cleaners that can turn a plumbing nightmare into a distant memory.
But here’s the magical part: when these two chemicals meet under the right circumstances, they don’t always go all Hulk-smash on each other. In fact, they can become quite… harmonious. It’s like a dramatic showdown that ends with a surprisingly peaceful resolution. And that resolution is what we call a balanced equation. Sounds a bit like balancing your checkbook, right? A little bit of this, a little bit of that, and everything adds up perfectly.
The Star Performers: Who Are They Anyway?
Let’s meet our main characters. First up, we have sulphuric acid. Imagine it as a very enthusiastic but sometimes too eager helper. It's a strong acid, meaning it loves to give away tiny particles called protons (don’t worry, we’re not going to get into nuclear physics here). Think of it like a really generous person who’s constantly handing out freebies. This eagerness is what makes it so useful in industry, breaking down things or kickstarting reactions.
Then there’s sodium hydroxide, often called lye or caustic soda. This one is a strong base. If sulphuric acid is a generous giver, sodium hydroxide is a really enthusiastic receiver, or maybe someone who loves to neutralize things. It’s the opposite of an acid. Imagine it like a calming influence, a chill friend who can talk down a heated argument. It’s incredibly useful for making soaps, paper, and yes, those drain cleaners I mentioned earlier.
When Opposites Attract: The Chemical Tango
So, what happens when this energetic giver (sulphuric acid) meets this calming receiver (sodium hydroxide)? It’s a classic case of opposites attracting! They come together in a chemical reaction, and it’s not a fight to the death. Instead, it's a beautiful dance of molecules.
Think of it like this: you have a bunch of kids who are super hyper and running around (the acidic properties of sulphuric acid), and then you have a group of really calm, organized teachers (the basic properties of sodium hydroxide). When these two groups are put together, the teachers help calm down the energetic kids. They interact, and what’s left is a much more peaceful, neutral group, and some new, useful things are formed.
The Magic Word: "Balanced"
Now, for the balanced equation part. This is where we get a little bit precise, like making sure you have exactly the right amount of flour for your cookies so they don't end up like hockey pucks or gooey messes. In chemistry, a balanced equation means that for every atom (the tiny building blocks of everything) that goes into the reaction, the exact same number of that type of atom comes out. Nothing is lost, and nothing magically appears. It’s all about conservation, like a really responsible accountant for atoms!
The chemical formula for sulphuric acid is H₂SO₄. That 'H₂' means there are two hydrogen atoms, the 'S' is one sulfur atom, and the 'O₄' means four oxygen atoms. Simple enough, right? Sodium hydroxide is NaOH, meaning one sodium atom, one oxygen atom, and one hydrogen atom.
When they react, they create sodium sulfate (Na₂SO₄) and water (H₂O). Sodium sulfate is a salt, and water is… well, it’s water! Essential for life, and also a pretty neutral substance. So, we’ve gone from a strong acid and a strong base to a salt and water.
Let's See It in Action (On Paper, Anyway!)
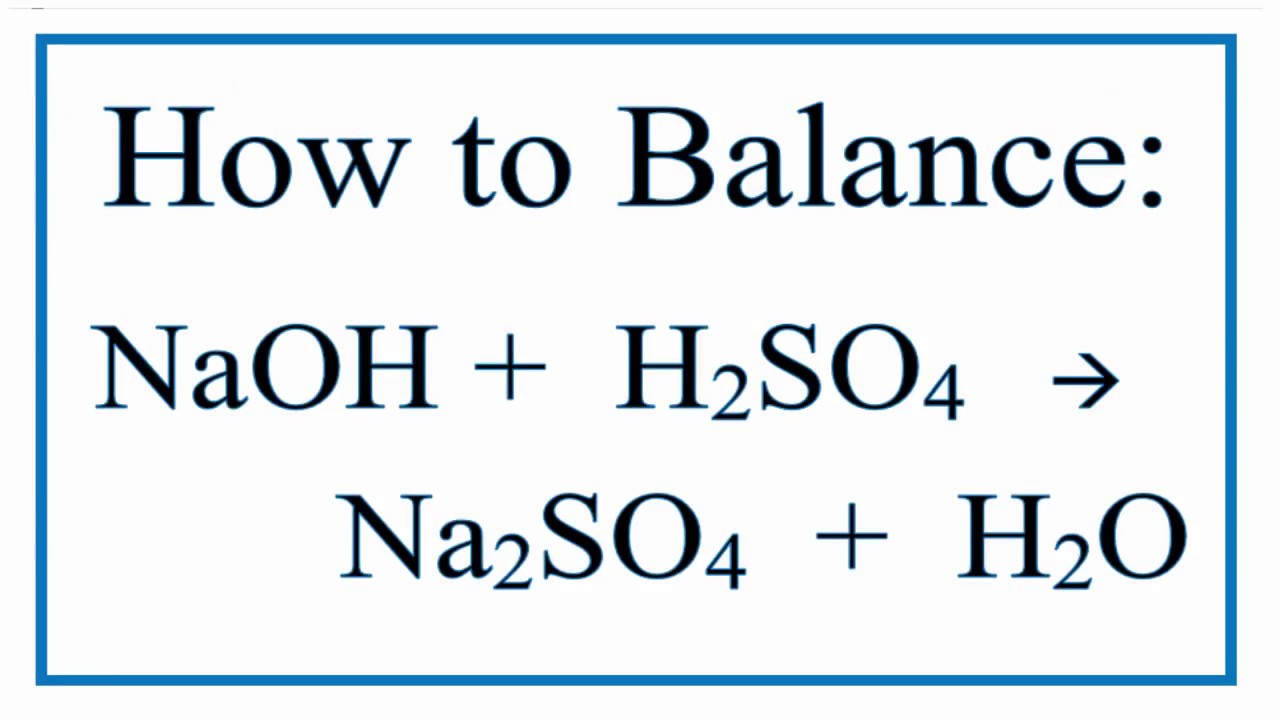
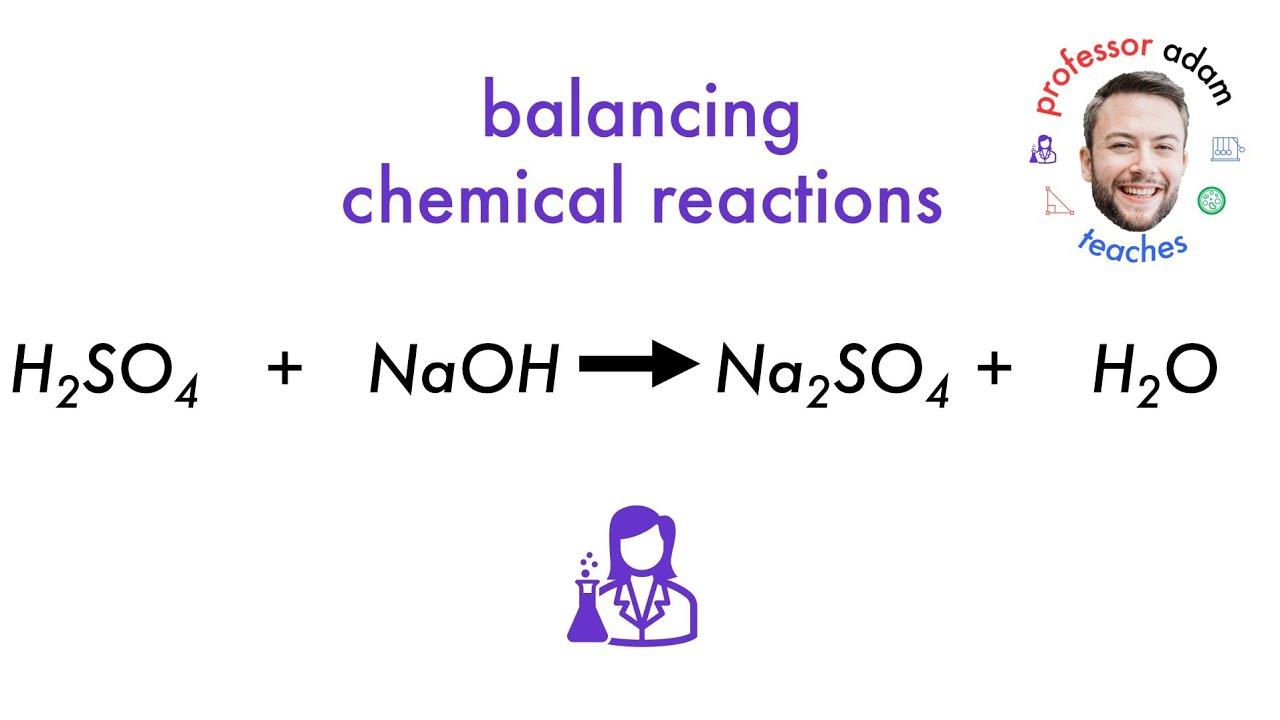
The unbalanced equation might look something like this: H₂SO₄ + NaOH → Na₂SO₄ + H₂O.
Now, let’s count our atoms on each side. On the left (what we start with): * Hydrogen: 2 from H₂SO₄ + 1 from NaOH = 3 * Sulfur: 1 * Oxygen: 4 from H₂SO₄ + 1 from NaOH = 5 * Sodium: 1
On the right (what we end up with): * Sodium: 2 * Sulfur: 1 * Oxygen: 4 from Na₂SO₄ + 1 from H₂O = 5 * Hydrogen: 2 from H₂O

Uh oh! See? Our hydrogen count is 3 on the left and 2 on the right. Our sodium count is 1 on the left and 2 on the right. It’s not balanced! It’s like trying to make a sandwich with one slice of bread and ending up with two. Not quite right.
The Balancing Act: Making it Perfect
To fix this, we need to add little numbers in front of the chemical formulas. These are called coefficients, and they tell us how many molecules of that substance are involved. We can’t change the internal makeup of the molecule (like turning H₂O into H₃O), but we can have more or fewer of them.
Let’s try putting a '2' in front of the sodium hydroxide (NaOH). Now we have: H₂SO₄ + 2NaOH → Na₂SO₄ + H₂O.
Let’s recount:
On the left:

- Hydrogen: 2 from H₂SO₄ + (2 * 1) from 2NaOH = 4
- Sulfur: 1
- Oxygen: 4 from H₂SO₄ + (2 * 1) from 2NaOH = 6
- Sodium: (2 * 1) from 2NaOH = 2
On the right:
- Sodium: 2
- Sulfur: 1
- Oxygen: 4 from Na₂SO₄ + 1 from H₂O = 5
- Hydrogen: 2 from H₂O
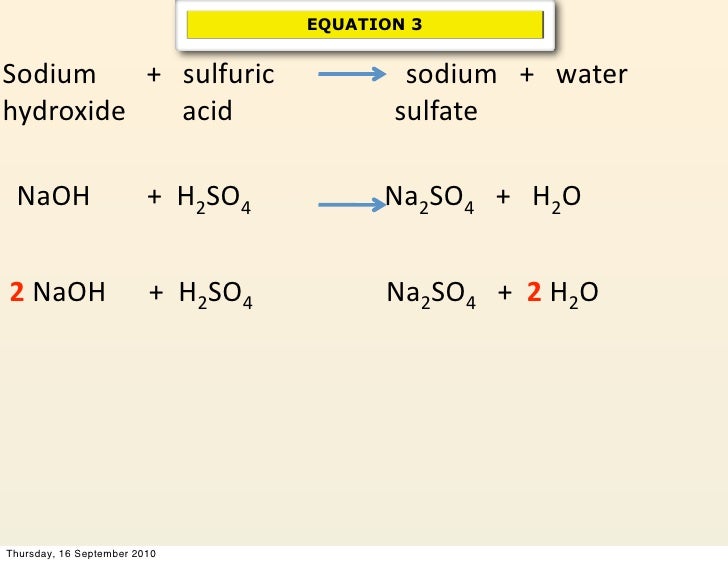
Still not perfect! We’ve fixed sodium, but now hydrogen and oxygen are off. Let’s try adding a '2' in front of the water (H₂O) on the right side. So, the equation becomes: H₂SO₄ + 2NaOH → Na₂SO₄ + 2H₂O.
Let’s do a final check:
On the left:
- Hydrogen: 2 from H₂SO₄ + (2 * 1) from 2NaOH = 4
- Sulfur: 1
- Oxygen: 4 from H₂SO₄ + (2 * 1) from 2NaOH = 6
- Sodium: (2 * 1) from 2NaOH = 2
On the right:
- Sodium: 2
- Sulfur: 1
- Oxygen: 4 from Na₂SO₄ + (2 * 1) from 2H₂O = 6
- Hydrogen: (2 * 2) from 2H₂O = 4
Ta-da! We have 4 hydrogens on both sides, 1 sulfur, 6 oxygens, and 2 sodiums. Everything matches up. The equation is finally balanced!
Why Should You Care? Because It's Everywhere!
Okay, so we’ve played chemist and balanced an equation. But why does this matter to you, the person just trying to enjoy a nice cup of coffee and avoid any accidental chemical spills?
Well, these reactions, or variations of them, are happening behind the scenes in so many places. Think about wastewater treatment. When sewage is processed, acids and bases are used to neutralize harmful substances, making the water safe enough to go back into our rivers and oceans. This balanced reaction is a small but crucial part of keeping our planet a little cleaner.
Or consider manufacturing. The production of fertilizers, dyes, and even some types of glass involve reactions with sulphuric acid. Sodium hydroxide is vital for making paper, textiles, and, as I mentioned, that glorious soap that gets you squeaky clean. When chemists and engineers design these processes, they absolutely need to work with balanced equations to ensure they’re using the right amounts of ingredients, minimizing waste, and making the process safe and efficient.
Even in your own home, while you’re probably not mixing concentrated sulphuric acid and sodium hydroxide (please, please don’t!), understanding the concept of balancing is like understanding how to follow a recipe. If you use too much baking soda or not enough egg, your cake is going to be… well, you know.
So, next time you see a product that promises to clean your drains or you hear about how we’re treating our water, spare a thought for the humble sulphuric acid and sodium hydroxide. They’re involved in a constant, fascinating, and often surprisingly gentle chemical dance. And that balanced equation? It's the neat, tidy, and utterly essential choreography that makes it all work, keeping our world running just a little bit smoother, one atom at a time.
