The Early Universe Contained Only One Element What Was It
Okay, so picture this: you're chilling on your couch, maybe scrolling through cat videos (no judgment, we've all been there), and you start wondering about... well, everything. Like, where did all this come from? Your couch, your phone, the snack you're currently devouring. It all feels so solid, so real. But what if I told you that for a really, really long time, the entire universe was way simpler? Like, ridiculously, almost boringly simple.
Imagine a toddler's crayon box with only one single crayon. That's pretty much the early universe. Not a lot of artistic freedom there, right? You can't exactly draw a rainbow with just one color. And that's exactly the cosmic conundrum we're diving into today. What was that one, solitary, foundational crayon that kickstarted everything we know and love (and also, you know, the stuff we don't love, like Mondays)?
It’s a question that tickles the brain cells, isn’t it? We live in a universe bursting with variety. We've got elements from the lightest, fluffiest hydrogen to the heavy, bling-y gold and everything in between. Stars are made of different things, planets are specks of cosmic dust, and we, well, we’re a rather complicated cocktail of many, many elements. So, how did we get from a single-note universe to this symphony of stuff?
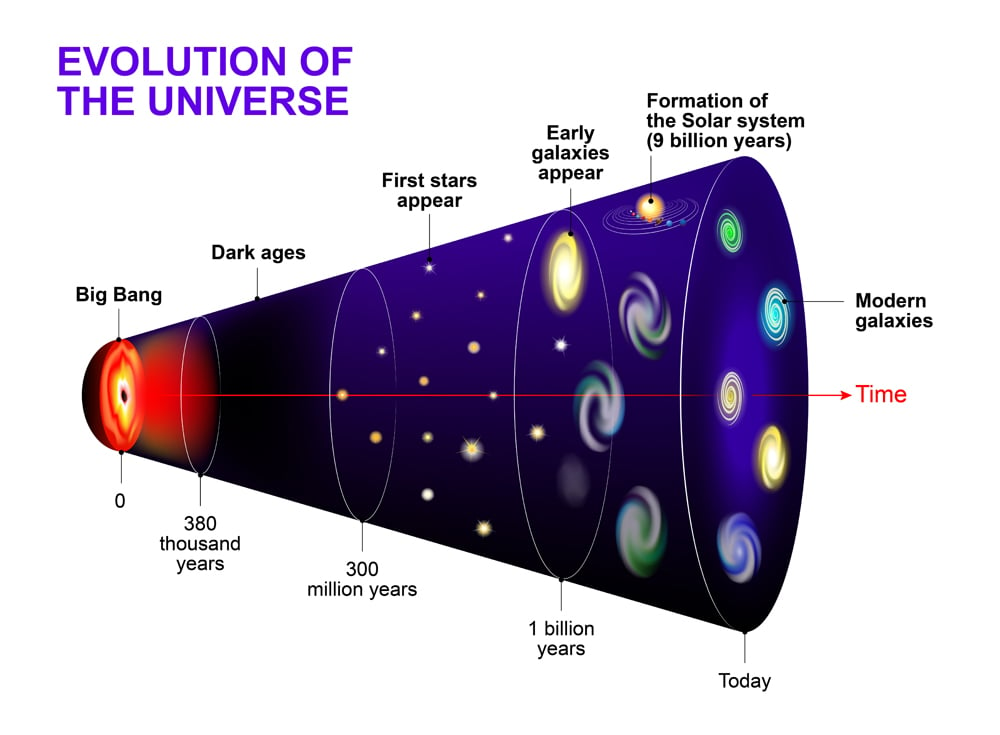
Let’s rewind, way, way back. Like, 13.8 billion years back. If you could time travel (which, sadly, is still a bit sci-fi, but a fun thought experiment, right?), you wouldn't see stars, or galaxies, or even atoms as we understand them. You'd see something… different. Something incredibly hot, dense, and surprisingly uniform.
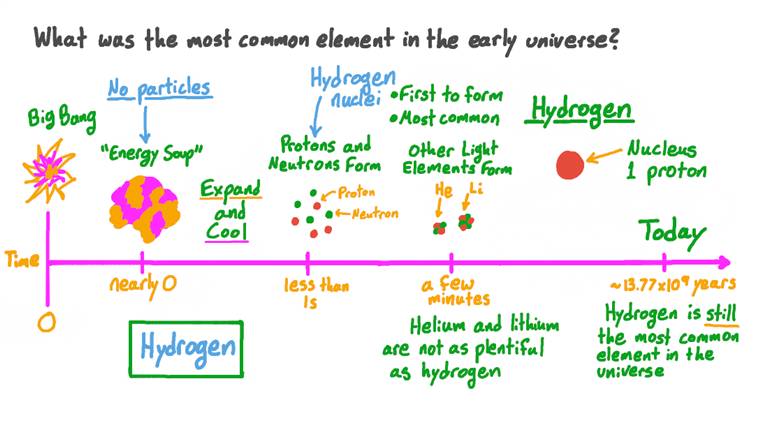
Think of it like a cosmic seed. A seed that contained the potential for everything, but was, in its earliest moments, just a single, fundamental ingredient. And that ingredient, my friends, was hydrogen. Yep, just good old hydrogen.
The Universe's First (and Only) Ingredient: Hydrogen!
I know, I know, it sounds almost… anticlimactic. You might have been expecting something more exotic, like pure starlight or a primordial blob of dark matter. But nope. The building block of the entire cosmos, at its inception, was the simplest element on the periodic table. Element number one, with just one proton in its nucleus and one electron buzzing around. Atomic number 1, baby!
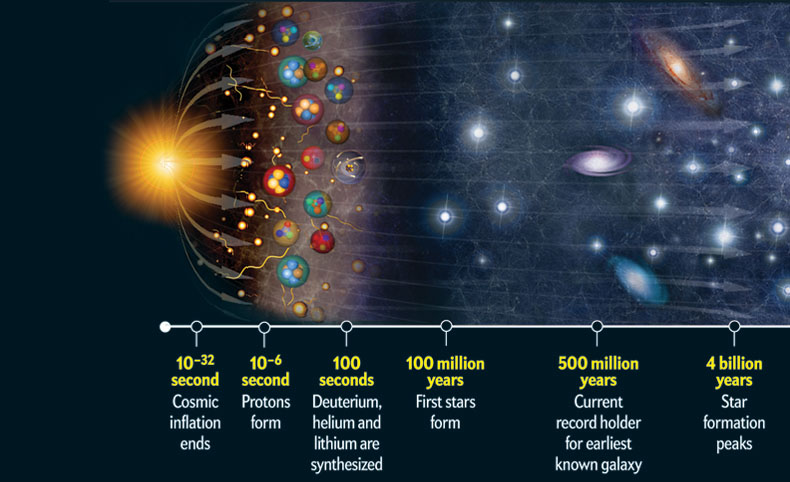
Why hydrogen? Well, it all comes down to the Big Bang. Now, the Big Bang wasn't an explosion in space, but rather the expansion of space itself. And in those first few microseconds, the universe was a super-energetic soup of fundamental particles. Protons and neutrons were zipping around, and electrons were free agents, not yet bound to anything.
As the universe expanded and cooled, things started to settle down a bit. And when the temperature dropped enough, those free protons and neutrons (which are made of even tinier things called quarks, but let's not get too deep into that rabbit hole unless you're armed with a PhD and a very strong cup of coffee) started to combine.
The easiest combination? A single proton and a single electron. Poof! You've got a hydrogen atom. And because protons and neutrons were the most stable things that could form at that stage, and they readily combined with electrons, hydrogen became the dominant, if not the only, element present in significant quantities.
It's a bit like when you're baking. You start with basic ingredients: flour, eggs, sugar. You can't make a cake without them. And for the universe, hydrogen was that essential, foundational ingredient. Without it, there'd be no stars, no planets, no us. Kind of mind-blowing when you think about it, right? All the amazing diversity we see is built upon this single, humble element.

So, Where Did the Other Elements Come From? The Cosmic Kitchen's Next Course!
If the universe started with just hydrogen, then how did we end up with oxygen in our air, iron in our blood, and silicon in our smartphones? This is where the real magic, and the really hot stuff, happens. The answer, my curious friends, lies within the fiery hearts of stars.
Stars are essentially giant, natural fusion reactors. And for billions of years after the Big Bang, all the stars that formed were made almost entirely of hydrogen. They were like cosmic furnaces, burning through their hydrogen fuel.
Inside the core of a star, the immense pressure and temperature force hydrogen atoms to fuse together. This process, called nuclear fusion, is what powers stars and makes them shine. The first step in this cosmic cooking process is fusing hydrogen atoms to create helium. Helium is element number two, with two protons in its nucleus. So, the universe started with hydrogen, and the first "new" element to be forged was helium, right inside stars!
But stars don't stop at helium. As they continue to fuse elements, they create heavier and heavier ones. Imagine a really, really big star. We're talking stars many times the mass of our Sun. These giants have enough gravity and internal heat to fuse helium into carbon. Then carbon can fuse into oxygen. Oxygen can fuse into neon, then magnesium, then silicon, and so on.

It’s like an elemental ladder, with each rung being a heavier element. The really heavy elements, like gold, silver, and uranium, are the hardest to make. They're forged in the most extreme stellar events: the explosive deaths of massive stars, known as supernovae. When a giant star collapses and explodes, the sheer energy released can fuse elements all the way up to iron and beyond. These explosions then scatter all these newly created elements back out into the cosmos.
And guess what? Those scattered elements, the "star stuff" from long-dead stars, become the raw material for new stars, new planets, and eventually, new life. So, every atom of carbon in your body, every atom of oxygen you breathe, was once inside a star. Pretty wild, right? We are literally made of stardust.
The Cosmic Cycle of Creation
This cycle of stars forming, fusing elements, and then dispersing them is fundamental to the universe's chemical evolution. The very first generation of stars would have been almost entirely hydrogen, with a tiny bit of helium. As they lived and died, they enriched the interstellar gas with helium and a smattering of heavier elements.
The next generation of stars formed from this enriched gas. These stars would have had a higher proportion of heavier elements (astronomers call these "metals," even though they're not just iron and steel – any element heavier than hydrogen and helium counts as a metal in this context). This process continued, with each generation of stars producing more and more of the heavier elements.
Our own Sun is a later-generation star, formed from a cloud of gas and dust that had already been seasoned by previous stellar generations. That's why it has a significant amount of heavier elements (like oxygen, carbon, and iron) that are necessary for forming rocky planets like Earth and, you know, for us to exist. If the Sun had formed from a cloud of only hydrogen and helium, we wouldn't be here having this chat.
So, the next time you look up at the night sky and see a twinkling star, remember that it’s not just a distant ball of gas. It’s a cosmic factory, a forge where the elements that make up you, me, and everything around us are being created. And that single, lonely element that started it all, hydrogen, is still the most abundant element in the universe, making up about 75% of its mass. It’s the fuel for almost all the stars and the foundation upon which all the cosmic complexity is built.
It's a beautiful, interconnected story, isn't it? From a universe of just one element to the incredible diversity we see today, all thanks to the relentless power and incredible artistry of stars. It's a reminder that even the simplest beginnings can lead to the most extraordinary outcomes. And that, my friends, is a truly stellar thought.
Think about it: that tiny, unassuming hydrogen atom, present in such abundance right at the beginning, set in motion a chain reaction of cosmic creation that ultimately led to you being able to read this right now. It’s like the ultimate butterfly effect, but with protons and electrons instead of flapping wings. Amazing!
