True Or False Diffusion Takes Place Only In Liquids

Let's talk about a science word. It’s called diffusion. You might think it only happens when you drop a teabag in hot water. You know, the color slowly spreading out. Or maybe when you spray perfume in a room and the smell fills everything. That’s diffusion in action.
But here’s a thought that might tickle your brain. What if diffusion isn’t just about liquids? What if it’s hiding in plain sight, all around us, even in places we don’t expect?
For a long time, the common wisdom was that diffusion was a liquid-only affair. Think of those science textbooks, all neat and tidy. Liquids: diffusion happens. Gases: diffusion happens. Solids: ...crickets.
It’s like saying your favorite song can only be heard on the radio. What about your headphones? What about that concert you went to? The music is still music, right?
So, let's playfully challenge this "only in liquids" idea. Is it really that simple? Or are we missing some sneaky, solid-based diffusion?
The Case of the Cozy Solids
Imagine you have a really old, comfy wooden table. Over the years, a tiny bit of the wood might have “migrated” into your sofa cushions. Or maybe, over eons, tiny particles from your favorite armchair have seeped into the rug.
This sounds a bit like science fiction, doesn't it? But bear with me. Think about what diffusion actually is. It's the movement of particles from an area where there are lots of them to an area where there are fewer of them. It’s like a crowd spreading out after a concert.
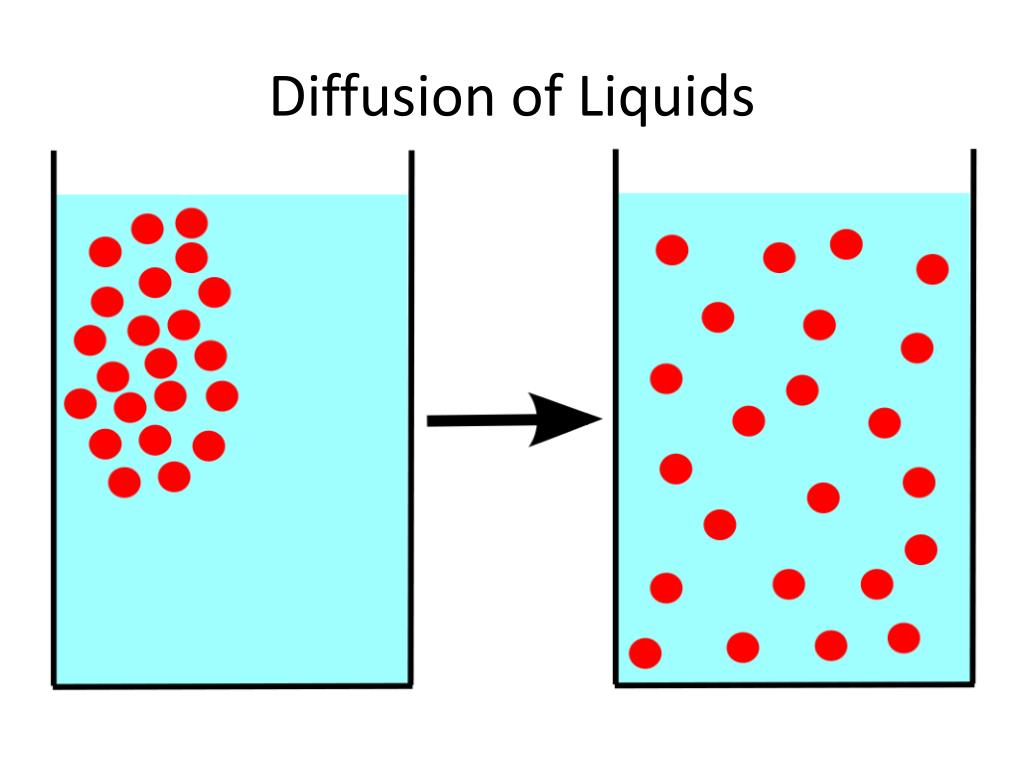
Now, solids can be quite crowded. But their particles are usually stuck in place, vibrating but not really moving around much. That’s the usual story, anyway.
However, even in solids, there are tiny spaces. And sometimes, just sometimes, a few adventurous particles can wiggle their way into those spaces. It’s not a stampede, it’s more like a slow, determined shuffle.
Metals: A Diffusion Love Story
Let’s talk about metals. When you heat up two different metals and press them together, something interesting can happen. Over time, the atoms from one metal can start to drift into the other metal. It’s like they’re getting to know each other.
This is actually a really important process in making things like alloys. Think of steel, which is a mix of iron and carbon. How do you think that happens? Diffusion plays a big role.

It’s not like you’re stirring them with a giant spoon. It’s a much more subtle, atom-by-atom mingling. They’re just… diffusing into each other. Slowly, patiently.
So, even though solids are usually thought of as being pretty solid (duh!), they can definitely have diffusion. It just takes a bit more oomph, or a lot more time. Or both!
The "Unpopular" Opinion
My “unpopular” opinion is this: diffusion isn't only a liquid thing. It’s more like a universal tendency for things to spread out. It just happens at wildly different speeds.
In liquids and gases, it's like a busy highway during rush hour. Things are moving, mixing, and mingling at a pretty good clip. You can see and smell it happening quickly.
In solids, it’s more like a sleepy village on a Sunday afternoon. Things can move, but they take their sweet time. You might not notice it unless you wait a very long time, or give them a bit of help.
Think of a tiny scratch on a metal surface. Over years, the edges of that scratch might actually smooth out a little. The metal atoms are diffusing to fill the void. It's subtle, but it's there.
Why Does This Matter (Besides Being Fun to Think About)?
Well, understanding diffusion in solids helps us in a bunch of practical ways. It’s crucial for making electronics work. It’s important for understanding how materials age and wear down. It even plays a role in how our bodies heal.
So, the next time you see a rusty nail or a worn-down stone, you can smile and think, "Ah, there's some slow-motion diffusion happening!" It’s not just a liquid phenomenon; it’s a fundamental force of nature.

It’s like the universe’s way of saying, “Everything eventually mixes and mingles, one way or another.” Whether it’s the smell of coffee spreading through your kitchen or the atoms of your favorite ring slowly rearranging over decades, diffusion is always doing its thing.
So, the next time someone tells you diffusion is only in liquids, you can wink and nod. You know the secret. You know about the cozy solids and the metal love stories. You know that diffusion is much, much more than just a wet affair.
It’s a quiet, persistent dance of particles, happening everywhere, even where you least expect it. And that, my friends, is a rather delightful thought.
Perhaps we've been a bit too narrow-minded about diffusion. It's time to give those solids a little credit for their patient mingling!
So, true or false? Diffusion takes place only in liquids? I’m leaning towards a playful, emphatic FALSE! It’s a much more widespread phenomenon, just with varying speeds and dramatic flair.
