What Ions Are Formed When Alkalis Dissolve In Water

Hey there, science buddies! Ever wonder what happens when those super reactive alkali metals, you know, the ones that practically scream when you throw them in water, decide to take a little dip? It's not just a fizz and a pop, oh no. It's a whole ionic party!
So, let's dive in. We're talking about the alkali metals. Think Lithium, Sodium, Potassium. The cool kids of the periodic table. They're all in Group 1. And they've got a serious need to share. Specifically, one electron. Just one little electron they're itching to get rid of. Like a toddler with a toy they’re bored with. And water? Water is the ultimate playmate.
The Big Splash: What's an Ion Anyway?
Before we get to the water, let's get our heads around what an ion is. Imagine an atom is like a balanced scale. It's got protons (positive charges) and electrons (negative charges), and they're perfectly matched. Everything’s chill.
But alkali metals? They have one extra electron hanging around. It’s like having a spare sock you never use. So, when they react with something, they’re like, "Here, you can have this!" And poof! They become an ion. They lose that negative electron, and suddenly, they have more positive protons than negative electrons. They're now positively charged. Bam! A positive ion. Or, as the chemists like to say, a cation. Fancy word, right?
Lithium's Little Dip
First up, let's meet Lithium. It's the smallest of the bunch, the lightweight champ. You toss a tiny bit of lithium into water, and it’s like a polite handshake. It’ll fizz a bit, maybe a tiny flame. Nothing too dramatic. Lithium is like the shy kid at the party.
What happens? That single, lonely electron on Lithium’s outer shell just can’t resist the pull of the water molecules. Water, you see, is a bit of a polar bear. It’s got a positive end and a negative end. And that’s super attractive to charged particles.
So, Lithium gives up its electron. It becomes a Lithium ion. It's now represented as Li+. See that little plus sign? That’s the badge of honor for losing an electron. It’s basically saying, “I’m positively charged and ready to mingle!” And the water molecules? They swoop in and surround this new Li+ ion, like a welcoming committee. It's called hydration. Pretty neat, huh?

Sodium's Big Entrance
Now, let's crank it up a notch. Meet Sodium. This is the one you probably see in videos. Drop a bit of sodium into water, and it’s a whole different story. We’re talking a significant fizz, a bit of a run-around on the surface, and often, a little blue flame. Sodium is the life of the party, but it can get a little wild.
Just like Lithium, Sodium has that one electron it’s just dying to shed. It’s a little heavier than Lithium, so it’s a bit more eager to react. When it hits the water, that electron is history. Zing!
Sodium becomes a Sodium ion. It's now Na+. That little plus sign again. This is a major player in our bodies, by the way. It helps nerve signals fire and keeps fluids balanced. So, while it’s exciting in a beaker, it’s pretty darn important in your own cells. Talk about a dual personality!
The water molecules can’t get enough of this Na+ ion. They surround it, stabilize it. The reaction is energetic, hence the heat and the flame. It’s a chemical fireworks show, all thanks to an ion having a bath.
Potassium's Fiery Farewell
And then there’s Potassium. Oh, Potassium. This one is the real drama queen. Drop Potassium into water, and you’re in for a show. It doesn’t just run around; it ignites with a spectacular purple flame. Think small, controlled explosions. It’s definitely not for beginners!
Potassium, being even bigger than Sodium, is even more eager to part with its electron. It’s like, “Here, take it! I don’t want it!” The reaction with water is incredibly vigorous. The heat produced is so intense that it actually ignites the hydrogen gas that's being released.
So, Potassium forms a Potassium ion: K+. Another happy, positively charged ion. This K+ ion is also vital for your body, especially for muscle and nerve function. It works hand-in-hand with Sodium. They’re like the dynamic duo of your biological world.
The energy released in the Potassium-water reaction is off the charts. It's a stark reminder of how much power is packed into these seemingly simple elements. It’s this energy that makes the hydrogen gas ignite, giving us that beautiful, and slightly terrifying, purple flame.
+an+alkali.jpg)
The Watery Embrace: Hydroxide Ions!
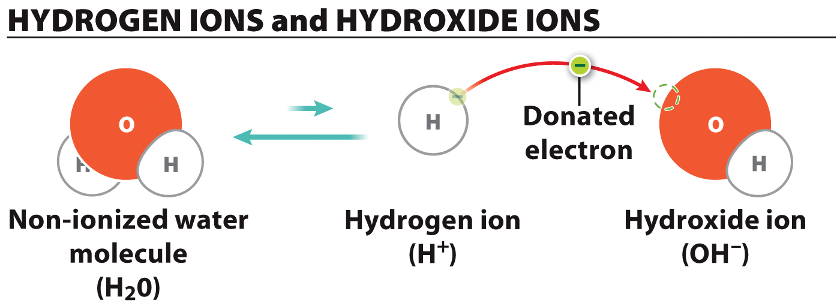
Now, here’s where it gets even more interesting. When alkalis dissolve in water, they don’t just form their positive ions. Oh no, the water itself gets involved in a bigger way. Remember how water molecules are polar? They have a slight negative charge on the oxygen atom and slight positive charges on the hydrogen atoms.
When an alkali metal, let's say Sodium (Na), reacts and forms a Sodium ion (Na+), it leaves behind a bit of a deficit in the solution. But the water molecules are clever. Some of them, when they're not too busy hugging the metal ions, can actually split up a tiny bit. You get some hydrogen ions (H+) and some hydroxide ions (OH-).
Here’s the kicker: the strong attraction between the positively charged alkali metal ion (like Na+) and the negatively charged hydroxide ion (OH-) is what truly makes these solutions alkaline. It’s the hydroxide ions that give the solution its characteristic basic or alkaline properties.
So, when Sodium dissolves, it’s not just Na+ floating around. It’s Na+ ions paired up with lots of OH- ions. This is why Sodium hydroxide (NaOH) is a strong base. It's essentially a boatload of Na+ and OH-.

Why Is This Fun?
Seriously, who wouldn't find this fun? We’re talking about elements that are so reactive they practically explode in water, creating ions that are essential for life. It’s like a mini-universe unfolding in your beaker.
Think about it: a single electron making all the difference. A tiny imbalance creating a charged particle. These ions then interact with water, changing its properties, creating bases that have countless uses, from making soap to cleaning products. It’s chemistry that’s both dramatic and deeply practical.
And the visuals! The fizzing, the running, the flames – it’s like nature’s own special effects department at work. It’s a reminder that even the smallest things, like an atom losing an electron, can have massive consequences.
So next time you see a science demonstration with an alkali metal, remember the ionic party happening. Remember the little metal atom saying, “Adios, electron!” and becoming a positively charged ion, ready to be hugged by water molecules and perhaps form a partnership with a hydroxide ion. It’s a wild, wonderful, and surprisingly important world of ions!
