What Is An Example Of A Giant Covalent Structure

Get ready for a mind-bending adventure into the world of giant covalent structures! Don't let the fancy name scare you; think of it as nature's way of building ridiculously strong, super-duper stable materials. We're talking about stuff so tough, it's like the superheroes of the chemical world, holding hands (or rather, sharing electrons) forever and ever!
Imagine a microscopic city where every atom is like a tiny builder, and instead of bricks, they're all constantly sharing tools (which are actually electrons). This sharing creates an unbreakable, interconnected network that stretches out, almost infinitely! It's like a massive, never-ending group hug between atoms.
So, what exactly is a giant covalent structure? Simply put, it's a humongous molecule where atoms are linked together by covalent bonds in a continuous, repeating pattern. There are no neat little individual molecules floating around; instead, you have one colossal, all-encompassing structure. It’s a bit like a sprawling city where every single building is fused to its neighbors, forming one massive, solid entity.
Think about it: most of the things we encounter are made of lots of small, separate pieces. Your LEGO castle is made of individual bricks. A bowl of cereal is made of countless little flakes. But a giant covalent structure is the opposite – it’s one enormous, indivisible unit at the molecular level. It’s the ultimate team player, where everyone is committed to the group!
Now, for the star of our show, the most famous and, dare I say, most awesome example of a giant covalent structure: diamond! Yes, that sparkly gem you might see on a fancy ring is a chemical marvel. It’s essentially a big, beautiful crystal of pure carbon atoms, arranged in a mind-bogglingly strong three-dimensional lattice.
Each carbon atom in diamond is bonded to four other carbon atoms. And these bonds? They're like superglue, but at the atomic level. They’re incredibly strong, sharing electrons so tightly that it’s almost impossible to break them apart. This is why diamond is the hardest natural substance on Earth!
Imagine trying to snap a spaghetti noodle. Easy peasy. Now imagine trying to snap a spaghetti noodle that’s been welded to a thousand other spaghetti noodles in every direction, with those welds being impossible to break. That’s kind of what it’s like trying to break diamond. It’s not just hard; it's ridiculously, astonishingly hard!

This incredible strength isn't just for show. It means diamond has some pretty amazing properties. For instance, it has a super high melting point. While your average ice cube melts at a measly 0°C (that's 32°F for our friends across the pond!), diamond needs temperatures hotter than the surface of the sun to even begin to melt.
And don't even think about trying to dissolve diamond in water. It's like asking a superhero to take a bath in a puddle; it just doesn't work. Giant covalent structures are generally insoluble in most common solvents because those solvents just aren't strong enough to break the bonds holding the whole thing together.
Because all the atoms are locked in this rigid, repeating structure, they don't have much freedom to move around. This lack of movement is why diamond is an electrical insulator. It’s like a traffic jam of electrons; they’re all tied up in those strong covalent bonds and can’t zip around to conduct electricity. So, while it’s pretty, you won't be using your diamond necklace to charge your phone anytime soon.
But wait, there's another carbon cousin that’s also a giant covalent structure and, in its own way, just as fascinating: graphite! You know, the "lead" in your pencils? Yep, that stuff. It looks very different from diamond, but it’s made of the exact same element: carbon!

The secret to graphite's difference lies in its structure. Instead of a 3D network, graphite is built in flat layers. Imagine a stack of incredibly thin, incredibly strong pancakes. Each pancake is a sheet of carbon atoms, and within each sheet, the carbon atoms are bonded very strongly to their neighbors, just like in diamond.
But the magic (or perhaps, the chemical engineering!) happens between these layers. The forces holding the layers together are much weaker than the bonds within the layers. Think of it like a deck of cards; the cards are strongly bonded to each other side-by-side, but the deck itself can easily be split into individual cards.
This is why graphite feels so slippery and is so soft. When you draw with a pencil, you're actually rubbing off these layers of graphite onto the paper. It’s like a controlled demolition, but on a microscopic scale! You can easily separate the layers, which is why graphite is used as a lubricant – it just slides right off.
And here's where graphite really shines (pun intended!): it's an excellent electrical conductor! Unlike diamond, where electrons are hog-tied in strong bonds, in graphite, some electrons are a bit more free. They can actually move around within those flat layers, carrying electrical current like tiny, speedy couriers.
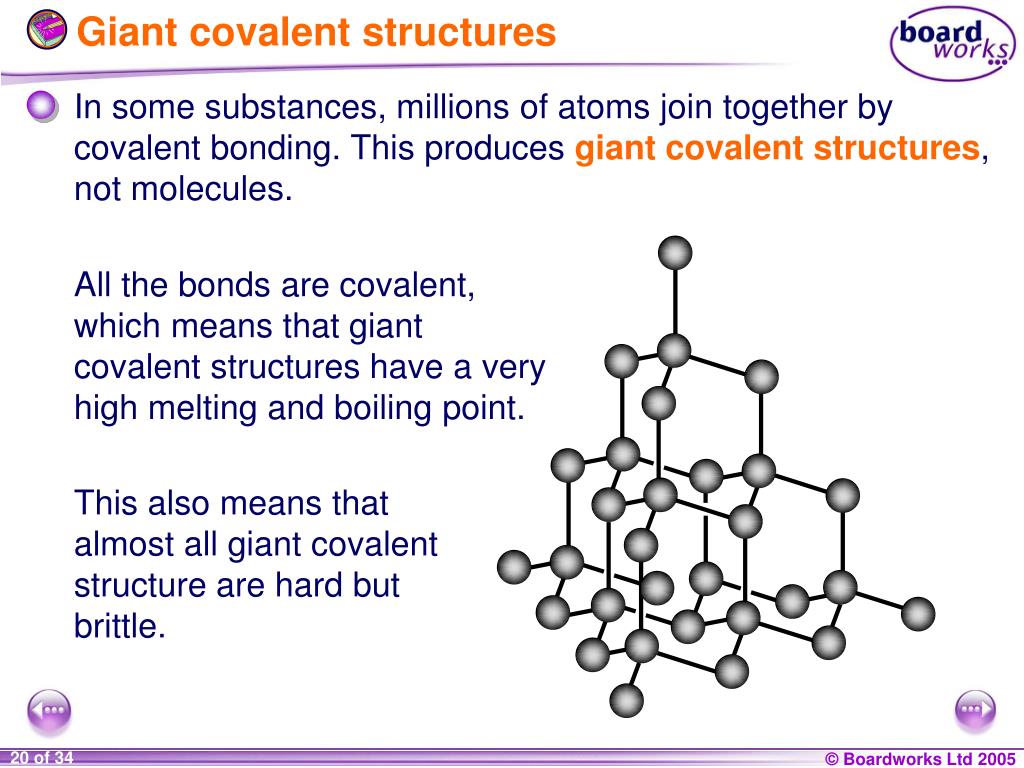
So, you have diamond, the super-hard, insulating marvel, and graphite, the slippery, conductive wonder, both made of pure carbon. It's a fantastic example of how the arrangement of atoms, not just the atoms themselves, can create vastly different properties. Nature is such a clever architect, isn't it?
Beyond carbon, there are other elements that can form these giant covalent structures. Take silicon dioxide, for example. You’ll find this common compound in sand and in the rocks that make up our planet. It’s also known as silica, and it forms a giant covalent network with silicon and oxygen atoms.
Think of quartz crystals; those beautiful, often clear structures are made of silicon dioxide arranged in a giant covalent lattice. Just like diamond, silicon dioxide is incredibly hard and has a very high melting point. It’s the backbone of many geological formations, silently enduring the pressures and temperatures of our planet.
Imagine a mountain range; its stability is, in part, thanks to the giant covalent structures within the rocks that form it. These structures provide the immense strength needed to withstand erosion, tectonic shifts, and the passage of eons. They’re the unsung heroes of geology, quietly holding the world together.

The properties of these giant covalent structures are what make them so useful. Their hardness makes them ideal for cutting tools and abrasives. Their high melting points mean they can withstand extreme temperatures, making them valuable in high-heat applications.
Their electrical insulating properties are crucial in electronics, where we need to prevent electricity from flowing where it shouldn't. And their electrical conductivity (in the case of graphite) is essential for things like electrodes in batteries and for creating conductive coatings.
So, the next time you admire a diamond, or use a pencil, or even just walk on a sandy beach, remember the incredible power and resilience of giant covalent structures. They are nature's way of building things that last, things that are incredibly strong, and things that perform amazing feats, all through the magic of shared electrons.
It's a testament to the elegant simplicity and profound complexity of chemistry. These structures, born from atoms holding hands (or rather, sharing electrons) in an eternal embrace, are fundamental to the world around us. They’re the silent workhorses, the unbreakable foundations, and the sparkling jewels of the material universe!
They remind us that even the smallest things, when organized in the right way, can create something truly monumental and awe-inspiring. So, let's give a virtual round of applause for these incredible giant covalent structures – the true titans of the molecular world!
