What Is The Difference Between Strong And Weak Acids

Let's talk about acids. Not the scary, "handle with extreme caution" kind you see in cartoons. Think more like the everyday, "oops, I spilled some lemon juice" kind. We all have a general idea of what an acid is. It's that sour taste, right? Like a really tart candy.
But did you know there are different levels of sourness? It's like comparing a mild sneeze to a full-blown, window-rattling cough. In the world of chemistry, we call these differences "strong" and "weak." And honestly, it's less about being a bully and more about personality.
Think of a strong acid as that friend who is always, and I mean always, ready to go. They dive headfirst into any situation. No hesitation. They're like a superhero with their cape flapping, ready to save the day. Or, you know, just really excited about whatever is happening.
When a strong acid meets water, it's like a complete and utter commitment. It's all in, no turning back. It breaks apart entirely, giving away all its little charged bits. We call these bits ions. They're like the acid's enthusiastic cheerleading squad, all separated and shouting their support.
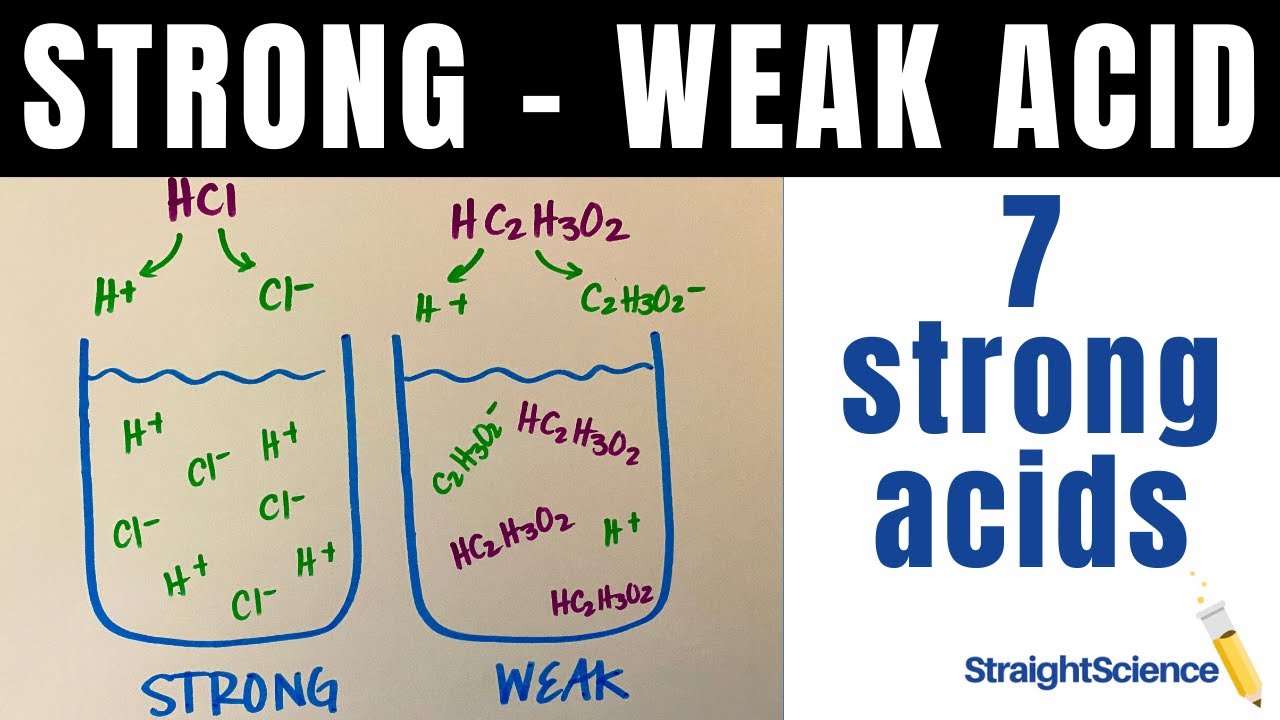
Imagine dropping a drop of hydrochloric acid (HCl) into water. Poof! It’s gone. All the H+ ions and Cl- ions are happily swimming around. They've completely dissociated. There’s no "maybe I'll break apart, maybe I won't." It's a definite yes.
This complete dedication means strong acids are really good at doing what acids do. They can be very reactive. They're the life of the chemical party, always sparking conversations and sometimes, a bit of a mess. They’re powerful.
Now, let's switch gears to weak acids. These are more like that friend who needs a bit of convincing. They're a little shy. They might dip a toe in the water but aren't always ready to jump all the way in. They're thoughtful.
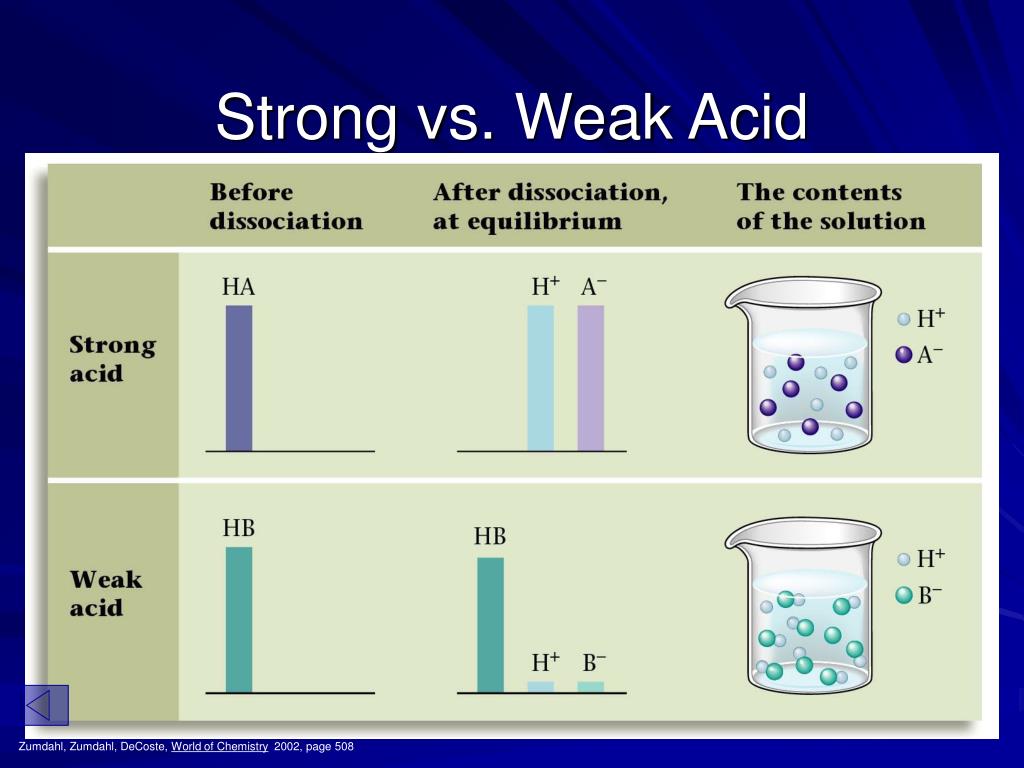
When a weak acid encounters water, it’s a more nuanced relationship. It only breaks apart partially. Some of the acid molecules will form those enthusiastic ion cheerleaders, but many will just hang out together, deciding not to split up just yet. It's a bit of a crowd.
Take acetic acid (CH3COOH) – that's the main ingredient in vinegar. When you add it to water, only a small percentage of those molecules will break into H+ and CH3COO- ions. The rest are still holding hands, enjoying each other's company. They’re not ready for a solo act.
This partial dissociation means weak acids aren't as dramatically reactive as their strong counterparts. They're more like a gentle nudge than a forceful shove. They can still get things done, but they do it with a bit more subtlety. They’re the quiet achievers.
So, the big difference boils down to this: strong acids are all-or-nothing. They completely break apart in water. Weak acids are more like, "let's see how this goes," and only partially break apart. It’s about their willingness to commit to becoming those charged ions.
Think of it like this: a strong acid is like a perfectly ripe strawberry. You bite into it, and you get the full, intense flavor. A weak acid is more like a slightly underripe banana. It's still got some sweetness, but it's not as explosive. You get hints of the flavor.
It’s also about how many free H+ ions are floating around. Strong acids create a whole parade of them. Weak acids create more of a small, polite gathering. This concentration of H+ ions is what makes something acidic. More H+ means a lower pH, which means more… well, acidity.
And this is where my unpopular opinion might come in. While chemists might get all excited about the technical differences in dissociation constants and equilibrium, I think we can appreciate the personalities of these acids. Strong acids are the go-getters, the ones who are always in your face with their chemical power.
Weak acids, on the other hand, are the patient ones. They’re the ones you can have a longer, more nuanced conversation with. They’re not going to overwhelm you with their intensity. They’re like a warm hug, not a firework display.
![Difference between Strong and Weak Acids [in Table Form] - Teachoo](https://cdn.teachoo.com/eb34c57a-aeb2-4e7c-aa0b-613a11a6595e/differentiate-between-strong-and-weak-acids-01.jpg)
It’s also interesting to note that "strong" doesn't always mean "dangerous" in the everyday sense. Sure, strong acids can be corrosive and require care. But so can some weak acids if you're not careful. It’s more about their chemical strength, their ability to donate protons.
For example, sulfuric acid (H2SO4) is a super strong acid. It's used in car batteries and is definitely something to be respectful of. But even a weak acid like citric acid, found in lemons, can have a noticeable effect. You wouldn't want to drink a whole glass of pure citric acid, would you?
So, while the chemistry books will tell you it's all about the equilibrium of dissociation, I like to think of it as their attitude. Strong acids have a "full send" attitude. Weak acids are more "let's take it slow." Both have their place. Both are fascinating in their own way.
We use strong acids for tough jobs, like cleaning drains or in industrial processes. They’re the heavy lifters. Weak acids are our everyday heroes, like in our food and our bodies. They’re the gentle facilitators. They help us digest food, for instance.

Consider carbonic acid (H2CO3). It’s formed when carbon dioxide dissolves in water. It’s weak, but it’s super important for regulating the pH of our blood. Without it, things would get messy. It’s the subtle bodyguard.
So, next time you encounter an acid, whether it's the sting of lemon juice or the power of a cleaning product, you can nod your head and think, "Ah, that's a personality thing." It's not about being good or bad, but about how completely they decide to unpack their chemical bags in water.
It's a spectrum, really. Some acids are just naturally more dramatic than others. They have a flair for the theatrical, a tendency to break apart with gusto. Others are more reserved, preferring to keep things a bit more contained.
And isn't that relatable? We all have friends who are the life of the party, ready to jump into anything. And we all have friends who are more introspective, who analyze before they act. Acids are no different. They just do it with protons and electrons.
So, while the science is important, let's not forget the charm. The bold, unwavering commitment of a strong acid. The gentle, thoughtful approach of a weak acid. They’re both essential, and they both bring their unique brand of acidity to the world. And that, I think, is something to smile about.
