When Metals React Do They Gain Or Lose Electrons

Ever wondered what makes your favorite jewelry sparkle or why some metals rust while others gleam forever? It all comes down to a microscopic dance of tiny particles called electrons! When metals get together, or interact with other substances, they’re not just passively sitting there. They’re actively engaging in a fascinating process of either giving away or snatching up these elusive electrons. It’s a bit like a high-stakes game of keep-away or a generous gift-giving ceremony happening at the atomic level. This fundamental principle is at the heart of so many everyday phenomena, from the batteries that power our gadgets to the protective coatings on our cars. Understanding whether metals gain or lose electrons isn't just for science buffs; it unlocks a deeper appreciation for the world around us, explaining why things behave the way they do and paving the way for amazing technological advancements.
The beauty of diving into the world of electron transfer lies in its sheer usefulness. Think about it: every time you charge your phone, flip a light switch, or even digest your food, the principles of electron transfer are at play. Understanding this process helps us design better batteries with longer lifespans, develop more efficient energy production methods, and even create new materials with incredible properties. For chemists and engineers, it's the bedrock of innovation. For us, the curious observers, it’s a peek behind the curtain of how the material world operates, turning the mundane into the marvelous. It’s the secret ingredient in everything from the vibrant colors in fireworks to the strength of steel bridges. This knowledge empowers us to understand and even manipulate the very building blocks of our universe.
The Great Electron Exchange: Who Gives and Who Takes?
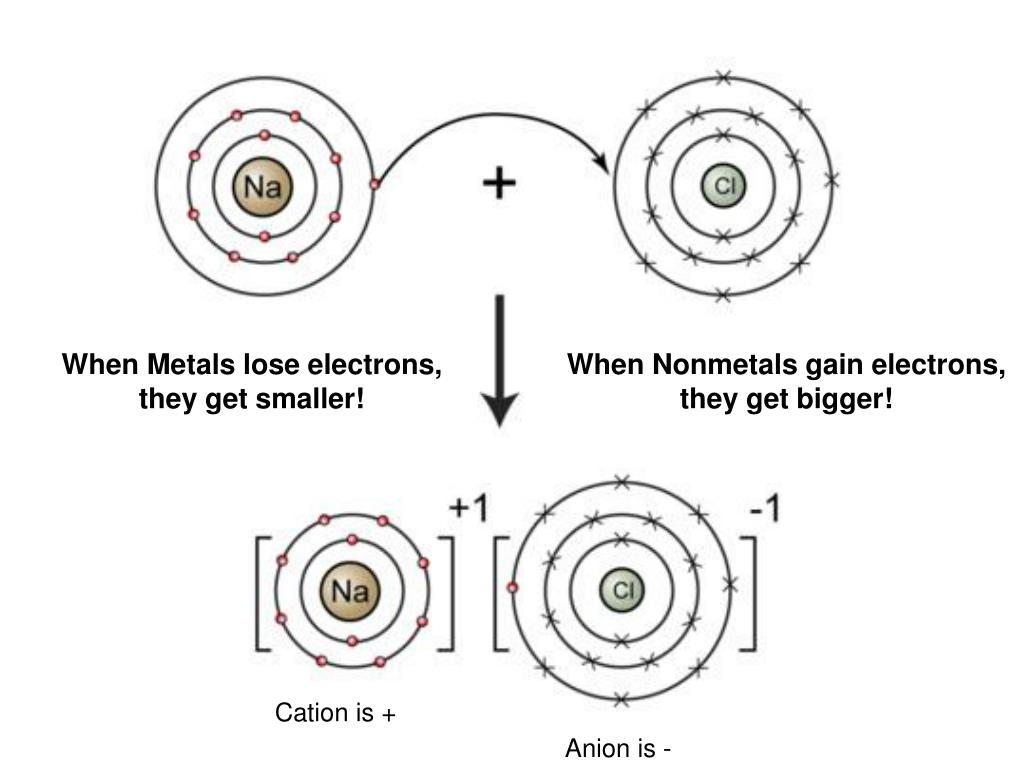
So, when metals engage in these electrifying reactions, do they end up with more electrons or fewer? The answer, in a nutshell, is that most metals tend to lose electrons. Imagine each metal atom as having a cozy arrangement of electrons orbiting its nucleus. Some of these electrons are held more tightly than others. Metals, generally speaking, have a relatively weak grip on their outermost electrons. They’re happy to let them go, especially when faced with an atom that’s eagerly waiting to snatch them up. This act of losing electrons is known as oxidation, and the metal that loses electrons becomes a positively charged ion. Think of it as the metal atom becoming a bit lighter, shedding some of its negative baggage and becoming a positive player in the chemical reaction.
Why are metals so keen to shed electrons? It’s all about achieving a more stable state. Just like we all strive for a comfortable balance in our lives, atoms constantly seek a state of lower energy, a kind of atomic harmony. By losing their outermost, less-bound electrons, metal atoms often achieve a more stable electron configuration, similar to that of noble gases, which are famously unreactive because they already have this perfect electron setup. So, for metals, losing electrons is often the path of least resistance, the quickest route to atomic contentment. This willingness to donate electrons is what makes metals such fantastic conductors of electricity and heat – those freed electrons can easily zip and zoom, carrying energy with them.

On the flip side, there are elements that are just as eager to gain electrons. These are typically nonmetals, like oxygen or chlorine. They have a strong attraction for electrons and are often found on the other side of these chemical reactions. When a metal loses an electron, something else needs to be there to catch it! This process of gaining electrons is called reduction. The element that accepts the electrons becomes a negatively charged ion. So, in a typical reaction between a metal and a nonmetal, the metal acts as the electron donor (gets oxidized) and the nonmetal acts as the electron acceptor (gets reduced). This give-and-take is fundamental to forming compounds and driving chemical changes.
Consider the common example of rust. When iron (a metal) is exposed to oxygen (a nonmetal) and water, the iron atoms readily lose electrons to the oxygen. The iron becomes positively charged iron ions, and the oxygen gains electrons, forming negatively charged oxide ions. These ions then combine to form iron oxide, which we know as rust. This is a classic case of a metal losing electrons to a more electronegative element. The shiny silver of your fork is iron, and the reddish-brown powder is its oxidized form, a testament to iron’s tendency to lose electrons.
Another fascinating area where this electron exchange is critical is in electrochemistry, the science of electricity and chemical reactions. Batteries, for instance, work by controlling the flow of electrons from one material to another through a circuit. In a typical battery, one electrode is made of a metal that readily loses electrons, while the other is a material that readily gains them. This difference in their electron-attracting or repelling tendencies creates an electrical potential, and when you connect the two electrodes with a wire, those electrons have a pathway to flow, generating electricity. So, the next time you power up your device, remember it’s a carefully orchestrated electron exchange, driven by metals losing and other substances gaining those tiny, energetic particles.
The concept isn't limited to just simple reactions. In more complex scenarios, metals might participate in a series of reactions, sometimes losing multiple electrons, depending on the conditions and what they're reacting with. However, the core principle remains: metals are generally electron donors. This inherent property is what defines their metallic character and makes them so indispensable in our modern world. From the robust structures that house us to the intricate circuits that connect us, the humble act of losing an electron by a metal is a powerful force shaping our reality.
