Which Giant Covalent Structure Is Used For Reinforcing Tennis Rackets
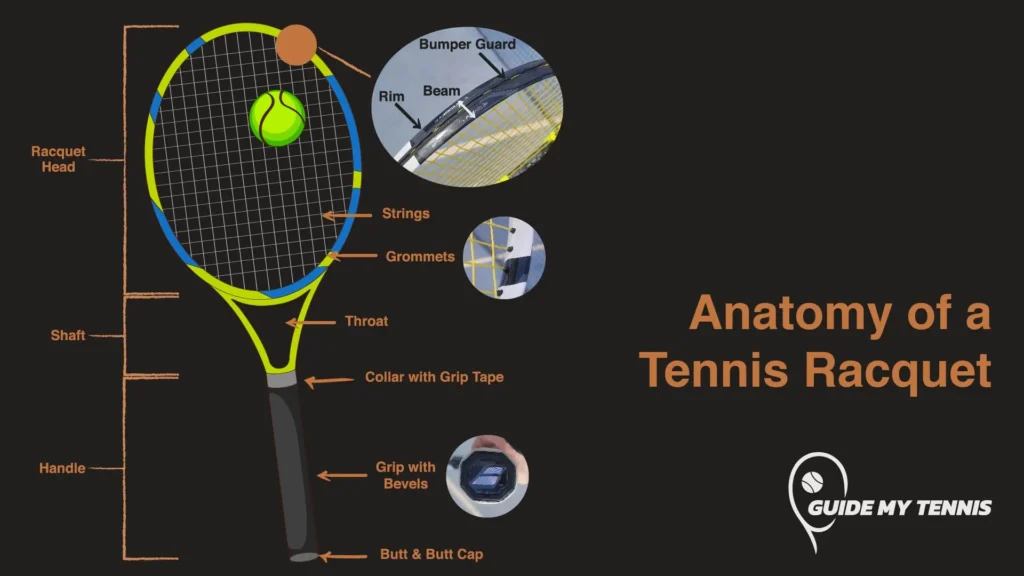
Hey there! Grab your coffee, settle in, because we’re about to dive into something seriously cool. Ever wonder what makes those tennis rackets so darn tough? I mean, they get whacked, they get slammed, they basically go through a war every match, right? And yet, they hold up. Pretty amazing, if you ask me. Well, there’s a secret weapon at play here, a tiny but mighty hero of the molecular world. And guess what? It’s a giant covalent structure. Yep, a giant one!
So, you're probably thinking, "Giant covalent structure? Sounds a bit… nerdy." And yeah, okay, maybe a little. But these things are the superheroes of the material world, seriously. They're like the bodybuilders of the chemical kingdom. They're huge, they're strong, and they don't break easily. Unlike, say, my New Year's resolutions. Those tend to… crumble. Ha!
Now, when we talk about giant covalent structures, we're not talking about tiny little molecules having a tea party. Oh no. We're talking about vast, interconnected networks of atoms. Think of it like a super-strong, unbreakable chain mail for molecules. Atoms are linked together, not just to a few buddies, but to loads of them, in a repeating pattern that stretches on and on. It’s like a never-ending LEGO fortress, but made of atoms. And that, my friends, is where the magic happens.
These structures are held together by covalent bonds. You know, those super strong “sharing is caring” bonds where atoms share electrons like they’re passing around the last slice of pizza. And when you have millions or even billions of these bonds all linked up in a giant, three-dimensional lattice? Boom. You’ve got yourself a structure that’s incredibly stable and, well, giant!
So, which of these molecular behemoths is the star of the show when it comes to reinforcing those tennis rackets? Drumroll please… it’s none other than diamond! Yep, the same stuff that makes your bling-bling sparkle and your fancy cutting tools do their thing. Diamond is basically the ultimate tough guy in the giant covalent family.
Think about diamond. It’s known for being one of the hardest substances on Earth. Seriously, you can’t scratch it easily. It’s ridiculously strong. And that’s because of its incredible structure. Each carbon atom in diamond is bonded to four other carbon atoms. And these bonds are arranged in a perfect tetrahedral shape. It’s like a tiny, perfectly built pyramid, and then those pyramids are all stacked together, perfectly interlocked. There are no weak points, no gaps, just pure, unadulterated atomic interlocking.

Imagine trying to break that. You’d have to break a lot of those super-strong covalent bonds to even make a dent. It’s like trying to dismantle a perfectly constructed brick wall by pulling out just one brick. You’re not going to get very far, are you? It’s this incredible network of strong covalent bonds that gives diamond its legendary toughness.
Now, you might be wondering, "Wait, are tennis rackets actually made of solid diamond?" And to that, I say, "Wouldn't that be something?!" A solid diamond tennis racket would probably cost more than my house. And let’s be honest, swinging that would feel like trying to hit a ball with a particularly aggressive paving stone. Ouch!
So, how does diamond help our humble tennis rackets? Well, it’s not usually in the form of giant, chunky diamonds. Instead, we're talking about something a little more… refined. We're talking about diamond nanoparticles. Or sometimes, diamond-like carbon (DLC). These are essentially super-tiny bits of diamond, or materials that have very similar, super-strong carbon-carbon bonding structures.

Think of it like this: you can’t build a whole house out of just one giant diamond (though imagine the bragging rights!). But you can use tiny, super-strong diamond fragments to reinforce the walls, make them tougher, more resistant to wear and tear. That's pretty much what's happening with tennis rackets.
These diamond nanoparticles or DLC coatings are added to the materials that make up the racket, like carbon fiber. Carbon fiber itself is pretty strong, you know? It’s made of long, thin strands of carbon atoms woven together. It’s already a pretty impressive material, used in everything from airplanes to fancy bicycles. But when you add these diamond-like bits? It’s like giving the carbon fiber a super-suit!
These tiny diamond particles get embedded within the carbon fiber matrix. They act like little microscopic reinforcements. When the racket gets hit by a fast-moving tennis ball, instead of the fibers flexing too much or even breaking, these diamond reinforcements help to distribute the stress. They act as tiny shock absorbers and incredibly rigid support beams, all at once. It’s like having a whole army of tiny, incredibly strong construction workers reinforcing the racket from the inside.
This makes the racket more stable. What does that mean for you, the player? Well, it means that when you hit the ball, the racket head doesn't twist or deform as much. It stays more solid. This leads to more consistent shots, better power transfer, and a more predictable feel. You know, that feeling of really connecting with the ball? Diamond reinforcements help with that. They help the racket be an extension of your arm, rather than something that wobbles around.
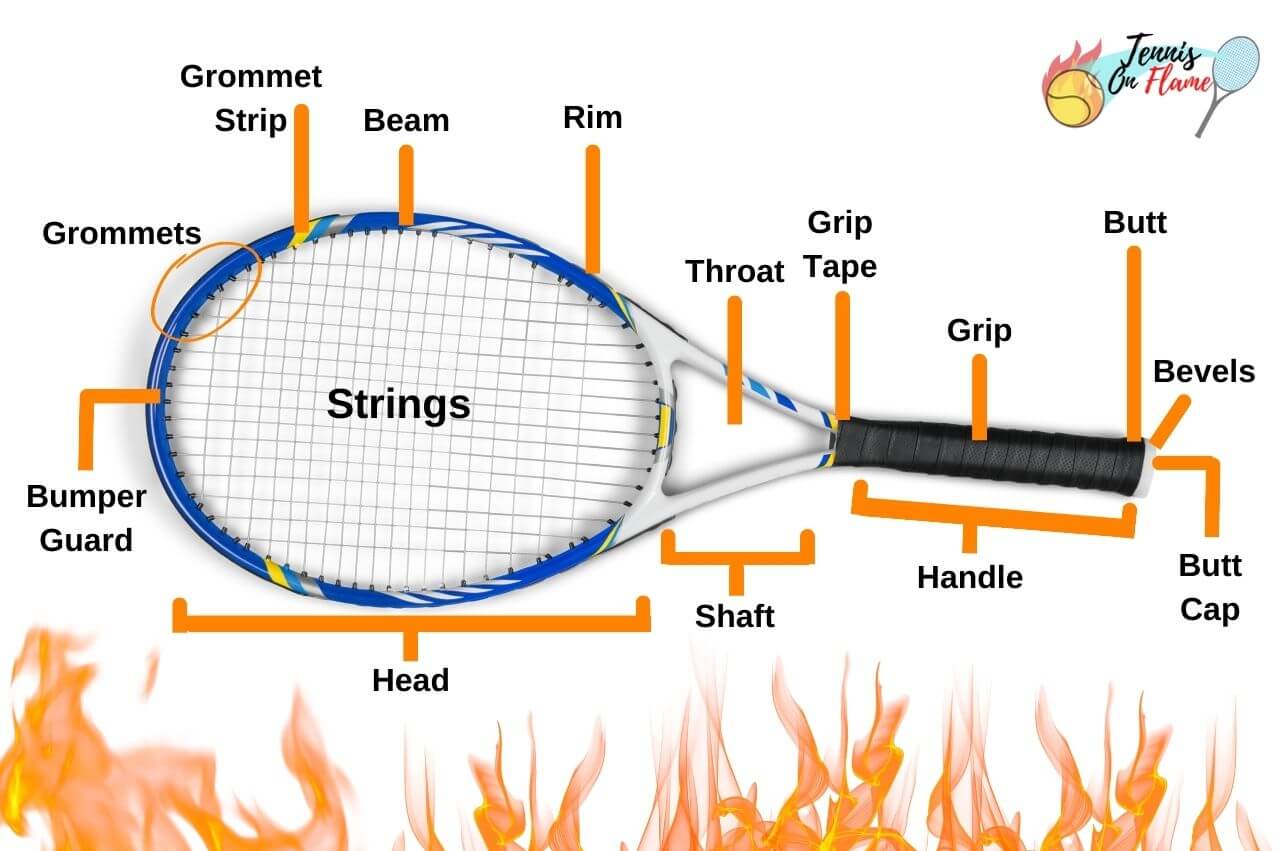
It also means the racket is more durable. Think about it. The constant impact, the vibrations, the occasional accidental thump against the ground (we've all been there, right?) – these can all take their toll on a racket. By adding diamond, you’re making the material much more resistant to wear and tear. It’s like giving your racket a protective shield. It’s less likely to get those annoying little nicks and scratches, and the overall structure is less prone to damage over time. So, your expensive racket lasts longer. Huzzah!
And then there’s the stiffness. While some players prefer a bit more flex in their racket for a softer feel, a certain level of stiffness is crucial for power. Diamond, being so inherently rigid, helps to increase the overall stiffness of the racket without adding excessive weight. It’s a delicate balance, of course. You don't want a racket that's so stiff it feels like you're hitting with a plank of wood. But these reinforcements allow for a controlled stiffness that translates into more powerful shots.
So, why diamond specifically? Well, it’s all about that pure carbon structure. Other giant covalent structures, like silicon dioxide (think quartz or sand), are also super strong. But diamond has that perfect, tight, interlocking arrangement of carbon atoms. Silicon dioxide, for instance, has oxygen atoms in there too, and while it's strong, it's not quite the same level of pure, unyielding strength as diamond.
And then there are other carbon structures, like graphite. Graphite is also a giant covalent structure, but its layers are held together by weaker forces. That's why it’s slippery and used in pencils. It’s strong in its own way, but not the kind of rigid, unbreaking strength you need for a tennis racket. Diamond is the undisputed king of hardness and rigidity when it comes to pure carbon forms.
The process of incorporating these diamond nanoparticles isn't exactly like sprinkling fairy dust. It’s a pretty sophisticated manufacturing process. They might be mixed into the resin that binds the carbon fibers together, or applied as a surface coating. The exact method depends on the racket manufacturer and the specific properties they're aiming for. But the end result is a material that’s significantly tougher and more resilient than plain old carbon fiber.
It's fascinating to think that something so tiny, something we can't even see with the naked eye, can have such a massive impact on the performance of our sports equipment. It’s a testament to the power of chemistry and material science. We're taking a material formed under extreme pressure deep within the Earth (or synthesized in labs) and using its incredible properties to help us hit a fuzzy yellow ball with more precision and power.
So, the next time you're watching a tennis match, or maybe even hitting a few balls yourself, give a little nod to the unsung hero: the giant covalent structure of diamond. It’s not just about shiny jewelry; it's about making our sporting dreams a little bit more robust. Pretty cool, huh? It makes you wonder what other everyday objects are secretly benefiting from these molecular superheroes. The possibilities are endless, really!
