Which Ions Positive Or Negative Will Be Oxidised During Electrolysis
Ever wondered what happens when you zap a liquid with electricity? It's not just about making pretty lights or charging your phone (though those are pretty cool too!). There's a whole hidden world of chemistry going on, and understanding it is like unlocking a secret superpower for your brain. Today, we're diving into the fascinating realm of electrolysis, specifically to uncover a little mystery: which ions, the positively charged or the negatively charged ones, are the ones that get oxidized. It’s a bit like a chemical detective story, and trust me, the solution is surprisingly straightforward and incredibly useful!
Electrolysis is a fantastic process that uses electrical energy to drive a non-spontaneous chemical reaction. Think of it as forcing a chemical change to happen that wouldn't normally occur on its own. It's the magic behind so many everyday marvels, from the shiny chrome plating on your car to the extraction of pure metals like aluminum and the production of chlorine gas, which is vital for everything from keeping swimming pools clean to manufacturing plastics. So, while it might sound a bit technical, the benefits are all around us, making our lives cleaner, more efficient, and even more aesthetically pleasing!
The Curious Case of Oxidation
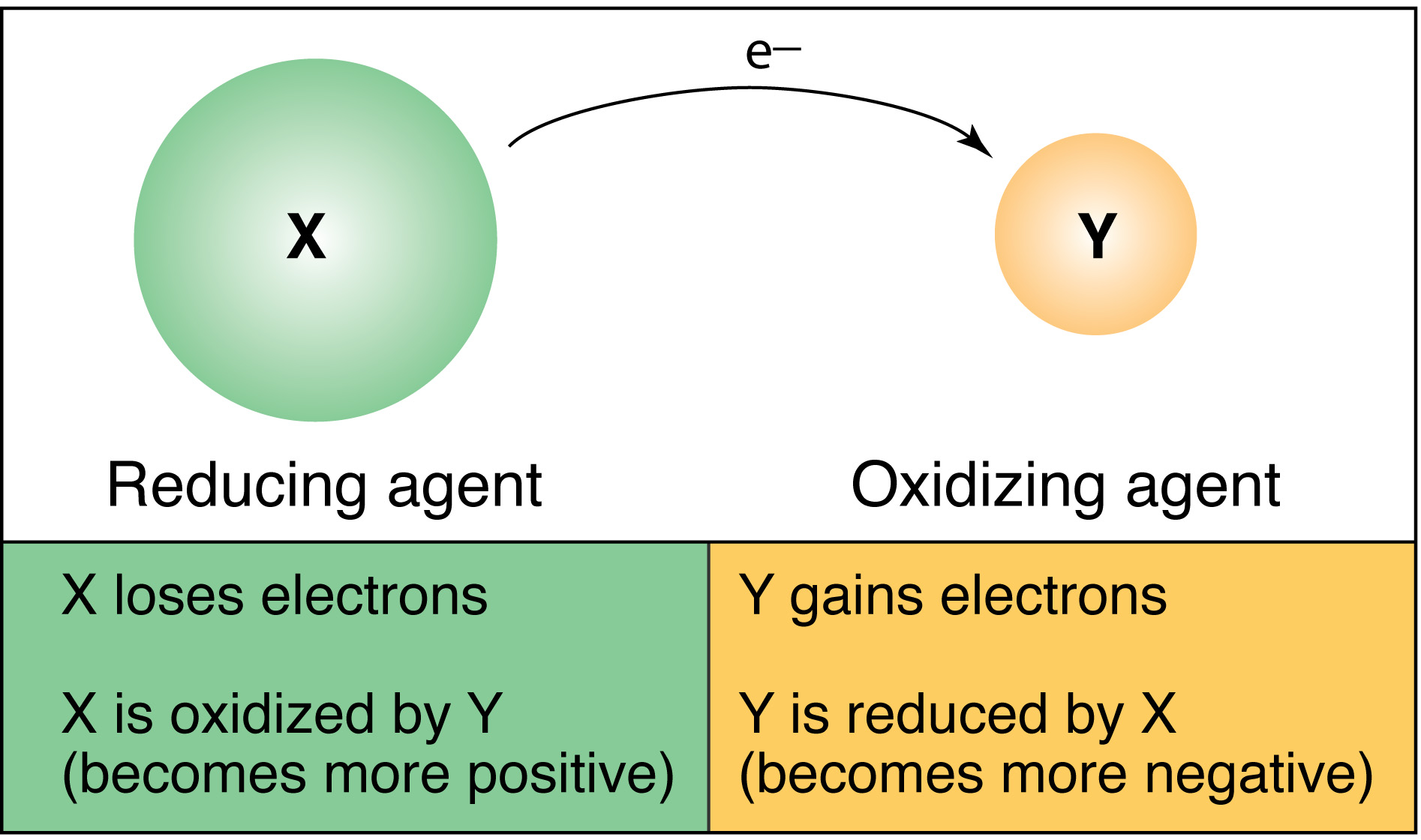
Before we get to the core question, let's quickly recap what oxidation means in this context. In the world of electrochemistry, oxidation is essentially the loss of electrons. Imagine a tiny particle, an ion, who has an extra electron (or more). During electrolysis, at a specific electrode, these ions will be nudged to give up those electrons. The opposite process, reduction, is the gain of electrons. Electrolysis involves both happening simultaneously at different electrodes.
Now, to the main event! In an electrolytic cell, we have two electrodes: the anode and the cathode. These are our chemical stages where all the action happens. The anode is connected to the positive terminal of the power supply, and the cathode is connected to the negative terminal. This electrical setup dictates which ions will be attracted to which electrode and, crucially, which will be oxidized.
The key to understanding which ions get oxidized lies in their charge and their position in the electrochemical series. It’s all about who’s more eager to lose electrons!
Chapter 8 Chemical Bonding - ppt download
So, which ions get oxidized? It's the anions, the negatively charged ions, that are heading for oxidation at the anode. Why? Because the anode is positively charged. Opposites attract, right? These anions, with their negative vibes (excess electrons), are drawn to the positive pull of the anode. Once they arrive, the conditions are ripe for them to let go of those electrons, thus undergoing oxidation.
Consider a simple example, like the electrolysis of molten sodium chloride (NaCl). In molten NaCl, we have positively charged sodium ions (Na⁺) and negatively charged chloride ions (Cl⁻). The Na⁺ ions, being positive, are attracted to the negative cathode. The Cl⁻ ions, being negative, are attracted to the positive anode. At the anode, the chloride ions (Cl⁻) are oxidized. Each Cl⁻ ion loses an electron to become a neutral chlorine atom (Cl). These neutral atoms then pair up to form diatomic chlorine molecules (Cl₂), which you might see bubbling away as a gas!

So, to sum it up in a super simple way: at the anode (the positive electrode), it’s the negative ions (anions) that will be oxidized because they are attracted to the positive charge and are more likely to lose their electrons.
It’s important to remember that the specific ions present in the electrolyte and their relative ease of oxidation or reduction play a role. Sometimes, water itself can be oxidized or reduced depending on the other ions present. However, the fundamental principle remains: at the anode, oxidation occurs, and it's typically the anions that are oxidized.
This concept might seem small, but it's a cornerstone of understanding how electrolysis works and how we can harness this power to create so many essential materials and processes. It’s a beautiful dance of charges and electron transfers, all driven by a simple electric current. Next time you see something shiny and metallic or use a product made possible by chemical manufacturing, you’ll have a little insight into the amazing world of electrolysis and the role of those eager-to-oxidize anions!

+than+electrons+(-)..jpg)