Which Of The Following Reacts Vigorously With Cold Water

Hey there, curious minds and home explorers! Ever find yourself staring at a bunch of household items, maybe after a bit too much doomscrolling or a particularly intense episode of that cooking show, and suddenly a question pops into your head? You know, the kind that’s both super specific and utterly intriguing? Today, we're diving into one of those head-scratchers: "Which of the following reacts vigorously with cold water?"
Now, before you imagine a mad scientist in a lab coat cackling over a bubbling beaker, let's chill. This isn't about dangerous experiments or anything that will turn your kitchen into a scene from a B-movie. It's more about understanding a bit of chemistry that, believe it or not, can subtly influence how we interact with the world around us. Think of it as a little mental refresh, like a cool sip of lemonade on a warm afternoon.
So, what are we talking about when we say "reacts vigorously"? We're not talking about a polite handshake. We mean a reaction that's noticeable, maybe even a little dramatic. We're talking about things like releasing heat, producing a gas, or even igniting. It's the kind of stuff that makes you go, "Whoa, that's pretty cool!"
The Usual Suspects (And Why They Don't Make the Cut)
Let's start by debunking a few common misconceptions. You might think of things you typically find around the house, and while some might be a little reactive, they’re generally pretty chill with H₂O.
Take, for instance, a good old bar of soap. Drop that into cold water, and what do you get? Lather! It dissolves, it gets slippery, and it does its job of cleaning. But vigorous? Nah. It’s more of a leisurely soak.
What about sugar? Similar story. It dissolves, sweetening your tea or coffee. It's a gentle process, no fireworks involved. Unless, of course, you're talking about the explosive potential of finely powdered sugar in the air, but that's a whole different ball game we’re not playing today.
And metals? Most common metals you’d find in your kitchen or workshop, like stainless steel, aluminum foil, or even a copper pot, are pretty stoic when it comes to cold water. They might rust over time (hello, iron!), or tarnish (looking at you, silver!), but it's a slow, evolutionary process, not a sudden outburst.
Even common cleaning agents, like bleach or vinegar, while they can react with other things, are generally safe to mix with plain old cold water. In fact, diluting them is often recommended! So, while they’re powerful, their interaction with water itself isn't the "vigorous" kind we're hunting for.
The Element of Surprise: Alkali Metals
Now, let's get to the real stars of our show. When we talk about elements that really make a splash with cold water, we’re usually looking at a specific group on the periodic table: the alkali metals. These guys are notorious for their energetic personalities.
You've probably heard of some of them, even if you didn't realize it. We're talking about elements like lithium (Li), sodium (Na), and potassium (K). These are all found in Group 1 of the periodic table, and they have one thing in common: they're incredibly eager to lose an electron. And water, with its slightly positive hydrogen atoms, is just the perfect partner for that electron exchange.
Lithium: The Mild-Mannered (Relatively) Cousin
Let's start with lithium. It’s the lightest of the alkali metals, and its reaction with cold water is the most… well, the most controlled of the bunch. Imagine a tiny fizzing sensation. You’ll see some hydrogen gas bubbles being released, and the lithium will move around on the surface of the water. It’s like a little dance.
Why is it the mildest? Because lithium atoms are smaller and have a stronger hold on their outer electron compared to their heavier alkali metal cousins. Still, it’s definitely a reaction, and it’s more energetic than your average household item.

Think of lithium in your phone or your electric car battery. That's a testament to its reactive nature, but in a controlled, electrochemical way. It’s a powerful element, but when it comes to water, it’s more of a spirited conversation than a full-blown argument.
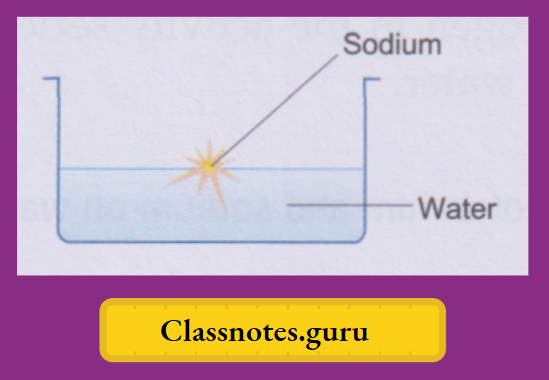
Sodium: The Classic Crowd-Pleaser
Ah, sodium. The element in table salt (sodium chloride). When pure sodium metal meets cold water, things start to heat up – literally. This reaction is a classic in chemistry demonstrations for a reason. You’ll see a noticeable fizzing, the release of hydrogen gas, and a good amount of heat generated. The sodium will often melt into a sphere and zip around the surface of the water, sometimes with a faint yellow flame (from the sodium vapor igniting).
This is because sodium atoms are larger than lithium atoms, making their outer electron easier to snatch away. The reaction is exothermic, meaning it releases heat. And that heat is enough to melt the sodium and even ignite the hydrogen gas being produced. It’s a bit like a tiny, contained fireworks display.
Culturally, sodium is everywhere. It's essential for life, and its compounds are vital. But pure sodium metal? That’s a different story. It’s stored under oil to prevent it from reacting with the moisture in the air. So, while you can't just pick up a piece of sodium from your spice rack, understanding its properties is pretty fascinating.

Potassium: The Unpredictable Powerhouse
Now, let's talk about potassium. If sodium is a spirited conversation, potassium is a full-blown, exuberant monologue. When potassium metal hits cold water, the reaction is significantly more vigorous than with sodium. The hydrogen gas produced ignites almost immediately with a loud 'pop' or even a small explosion. This is due to the greater amount of heat generated by the reaction, which easily ignites the hydrogen.
Potassium atoms are even larger than sodium atoms, and their outer electron is even more loosely held. This makes them even more reactive with water. It’s a beautiful, albeit slightly terrifying, display of elemental energy. This is the kind of reaction that commands your full attention.
Potassium is also vital for life, found in bananas and potatoes, but again, the pure metal is a different beast. It's so reactive that it's rarely encountered outside of specialized laboratories. It’s a reminder that even the things that sustain us can be incredibly powerful in their elemental form.
Why Does This Matter to Us?
Okay, so we've established that lithium, sodium, and potassium are the main players when it comes to vigorous reactions with cold water. But why should you care? It’s not like you’re going to be conducting alkali metal experiments in your bathtub.
Well, it’s about appreciating the incredible diversity and power of nature. It’s about understanding that even the most commonplace substance, like water, can behave in wildly different ways depending on what it interacts with. It’s a peek behind the curtain of how the world works.

Think about it. Water, that seemingly simple, clear liquid. It can be a gentle cleanser, a life-giving force, or, in the presence of certain elements, a catalyst for dramatic chemical change. It’s a reminder of the hidden potential in everything.
Practical (and Fun!) Takeaways
While you shouldn't be messing around with alkali metals, this knowledge can inform some everyday observations and even add a bit of flair to your conversations.
- Safety First (Always!): This is the most important takeaway. Never experiment with reactive substances. If you ever encounter something you're unsure about, err on the side of caution and keep it away from water. Especially if it looks like a shiny, soft metal that might be stored in oil.
- Appreciating Your Salt: Next time you're salting your food, remember that the sodium component is an element that, in its pure form, is incredibly reactive with water. It's a great conversation starter at the dinner table! "Did you know this salt is made from an element that can practically explode in water?"
- Science in Pop Culture: You might have seen depictions of chemical reactions in movies or TV shows. Understanding these basic principles helps you appreciate the (sometimes exaggerated!) science being presented. It's like having a secret decoder ring for nerdy cool moments.
- The Periodic Table is Awesome: The periodic table isn't just a chart for chemists. It's a map of the building blocks of our universe. Learning about elements like the alkali metals gives you a glimpse into their unique personalities and behaviors.
A Moment of Reflection
As I wrap this up, I’m reminded of how much complexity lies beneath the surface of our seemingly ordinary lives. We interact with water every single day – we drink it, we bathe in it, we use it to clean. It's so ubiquitous that we often take its properties for granted.
But then you learn about a tiny piece of metal, like sodium, that can cause water to fizz, melt, and even ignite. It’s a stark contrast, isn’t it? It speaks to the idea that even the most familiar things have hidden depths, and that there’s always more to discover if we just take a moment to ask "why?" or "what if?"
So, the next time you’re enjoying a glass of cool water, take a second to appreciate its calm demeanor. And maybe, just maybe, spare a thought for its more excitable alkali metal cousins, and the fascinating, energetic dance they perform when they meet.
