Why Do Giant Covalent Structures Have High Melting Points

Okay, let’s talk about those rock-solid types in the chemistry world. You know the ones. They’re the ultimate commitment-phobes when it comes to melting. We’re talking about giant covalent structures. You might have heard of them. They’re the ones that make you think, "Seriously? You're not even budging?"
Imagine you’re trying to break up a really, really big party. Not just a few friends chilling, but like, the entire neighborhood has decided to have a potluck that lasts for days. And everyone is holding hands. Like, everyone. That’s kind of what’s going on inside a giant covalent structure. It’s not just a few molecules having a quick chat. Nope. It’s a massive, interconnected network where atoms are all holding onto each other with super strong, covalent bonds. It’s like they’ve all agreed to be best friends forever, and they’re not letting go of anyone.
Think about it. When you want something to melt, you’re essentially asking it to relax a bit, to loosen up its grip, to maybe take a break from all that intense togetherness. You’re trying to get the little bits to slide past each other. But with these giant covalent structures, there are no little bits that can easily slide. It’s one giant, happy (or perhaps intensely dutiful) family. To get them to melt, you’d have to, like, break off a significant chunk of this super-gluey connection. And that, my friends, takes a whole lot of effort. A whole lot of heat. A whole lot of oomph.
It's like trying to convince a toddler to share their most prized toy. They're not just going to hand it over. They're going to hold on with all their might. And that’s what these atoms are doing. They’ve got these powerful covalent bonds, which are basically the ultimate handshake. They’re sharing electrons, and that’s a pretty serious commitment. It’s not like a weak ionic bond where it’s more of a polite nod. Nope, this is a full-on, electron-sharing hug that’s incredibly difficult to pull apart.

So, when we talk about high melting points, we’re not talking about a little bit of warmth making things a bit gooey. We’re talking about temperatures that would make your oven weep. We’re talking about heat levels that could make a dragon sweat. It’s because you’re not just warming up a few individual molecules. You’re trying to inject enough energy to disrupt this massive, interconnected network. You’re trying to break those strong, covalent bonds that hold the whole thing together. And breaking covalent bonds is hard work, people! It’s like trying to untangle a ball of yarn that’s been knitted by a thousand busy squirrels. It’s going to take some serious patience and probably a bit of frustration.
Let’s take some of the usual suspects. You’ve heard of diamond, right? That sparkly thing that makes people go “oooh” and “aaah”. Diamond is a giant covalent structure. And that’s why it’s so ridiculously hard and has a melting point that’s off the charts. It’s basically a perfectly structured crystal of carbon atoms, all linked together like a super-secure chain-mail suit. You try melting that! You’d need more heat than a supernova. And then there’s silicon dioxide, otherwise known as sand. Yep, that stuff you build sandcastles with (until the tide comes in, of course). Sand is also a giant covalent structure. It’s silicon and oxygen atoms all holding hands in a repeating pattern. It's why glass, which is made from silicon dioxide, is so tough and needs such high temperatures to be molded. It’s not just some flimsy powder; it’s a united front!
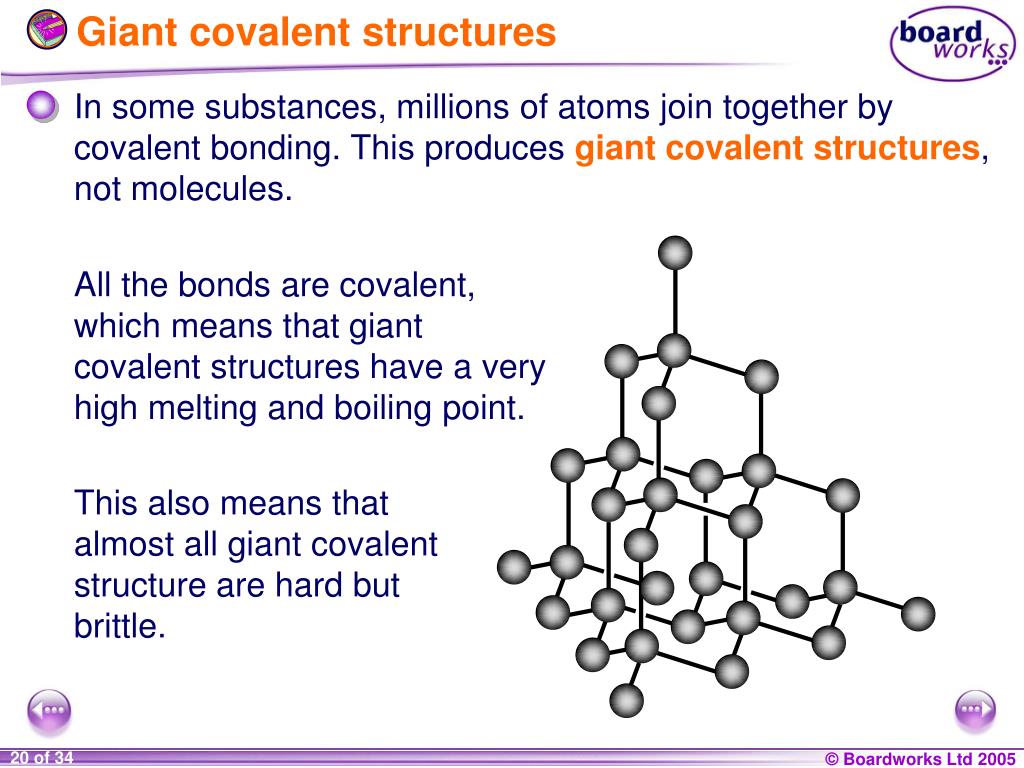
"It's like they've all agreed to be best friends forever, and they’re not letting go of anyone."
It’s this incredible strength of the covalent bonds, repeated over and over and over again in a huge, three-dimensional network, that gives these substances their incredibly high melting points. They're not just melting a few particles; they're trying to break the very fabric of the structure. It requires a monumental amount of energy to overcome those forces. So, the next time you encounter a substance with an absurdly high melting point and discover it’s a giant covalent structure, just remember: you’re dealing with a material that’s taken commitment to a whole new level. They’re the ultimate, unshakeable squad. And frankly, I kind of admire their dedication. Even if it makes lab work a bit more challenging.
It’s the chemical equivalent of saying, "I'm not going anywhere, and neither are you." And that, my friends, is a beautiful, albeit stubborn, thing.
