Why Is Cryolite Used In The Extraction Of Aluminium

Ever found yourself admiring the sleek lines of your smartphone, the sturdy frame of your bicycle, or even the effortless glide of an airplane wing? Chances are, you’ve been enjoying the magic of aluminum. This incredibly versatile metal is everywhere, from the foil wrapping your lunch to the pots and pans that grace your kitchen. But have you ever stopped to wonder how this ubiquitous metal actually gets from the earth to your hands? It’s a journey that involves a rather unusual, almost mystical-sounding ingredient: cryolite.
Now, before you start picturing a cryo-chamber for superheroes, let’s demystify this. Cryolite, or sodium hexafluoroaluminate to its friends, is a mineral. Think of it as a special kind of rock, but one with a superpower when it comes to aluminum extraction. And while it might not have the glitz and glamour of, say, a diamond, its role in our modern world is nothing short of spectacular.
So, why is this particular mineral so crucial in the grand scheme of aluminum production? Well, to put it simply, aluminum, in its rawest form, is incredibly stubborn. It’s locked away in a very stable compound called alumina (aluminum oxide). Imagine it as being in a super-secure vault, and we need a special key to get it out.
The Alumina Challenge: A Locked Vault
Aluminum oxide, the source of all our beloved aluminum, is found in a mineral called bauxite. Bauxite is dug from the ground, and then it's processed to create pure alumina. This alumina looks a bit like fine, white powder. It’s pure, it’s ready, but it’s also incredibly resistant to being broken down. Melting pure alumina directly would require temperatures so astronomically high that it would be practically impossible and wildly expensive to achieve on an industrial scale. We’re talking hotter than the surface of the sun, and nobody wants to deal with that kind of electricity bill!
This is where our star, cryolite, steps in. It’s not just a supporting actor; it’s the co-star that makes the whole production possible. Without cryolite, the industrial extraction of aluminum, a process we now take for granted, would simply not be feasible. It’s the secret sauce, the magic ingredient that lowers the temperature and makes the whole operation work smoothly.
Enter Cryolite: The Temperature Tamer
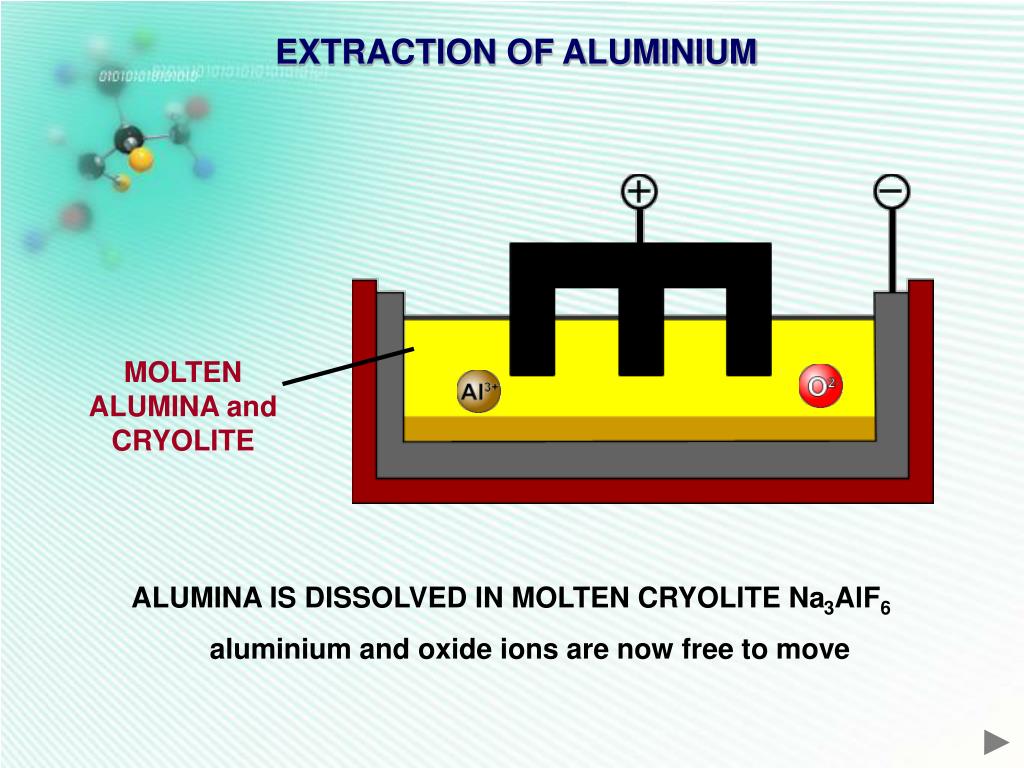
The process used to extract aluminum from alumina is called the Hall-Héroult process, named after its inventors, Charles Martin Hall and Paul Héroult, who independently discovered it in 1886. It’s a marvel of industrial chemistry, and cryolite is at its heart. This process involves dissolving alumina in molten cryolite. This creates a bath that conducts electricity very well, even at much lower temperatures than alumina alone would require.
Think of it like this: alumina is a brick wall. Trying to break it down directly is like trying to punch through it. Cryolite, when mixed with alumina and heated, creates a sort of "solvent" or "carrier fluid" for the alumina. This molten cryolite bath acts like a warm, welcoming swimming pool where the alumina can finally relax and be broken apart. The electricity then does the heavy lifting, essentially "pulling" the aluminum atoms away from the oxygen atoms.
The beauty of cryolite here is that it dramatically lowers the melting point of the alumina. Instead of needing temperatures reaching 2000 degrees Celsius (that’s over 3600 degrees Fahrenheit!), the molten cryolite bath allows the process to happen around 950-970 degrees Celsius (about 1740-1780 degrees Fahrenheit). This is still very hot, mind you – hotter than any oven you’ve ever used! – but it’s a temperature that’s achievable and manageable with modern industrial technology.

Furthermore, cryolite is incredibly stable in this molten state. It doesn't break down easily, meaning it can be reused over and over again. This makes the process much more efficient and economical. It’s like having a super-durable, reusable tool that keeps working perfectly, batch after batch.
Fun Fact: For a long time, the only significant natural deposit of cryolite was found in Ivittuut, Greenland. This made Greenland a crucial player in the early days of aluminum production. Imagine the buzz around this remote, icy location! It was like the Klondike Gold Rush, but for a frosty, white mineral.
The Chemical Tango: A Deeper Dive
Let’s peek under the hood just a little. When alumina (Al₂O₃) is dissolved in molten cryolite (Na₃AlF₆), it forms a complex mixture. The electricity, supplied by large carbon anodes, then acts as the breaking force. Oxygen atoms are attracted to the anodes and react with the carbon, forming carbon dioxide gas, which bubbles away. The aluminum atoms, freed from their oxygen partners, are then attracted to the cathode (usually the lining of the electrolysis cell) and collect as molten aluminum.
So, cryolite isn't chemically consumed in the way a reactant might be. Instead, it acts as a medium. It facilitates the reaction by providing a conductive, low-melting-point environment. It’s the ultimate enabler, allowing the magic of electrochemistry to do its work efficiently.
The chemical reaction that’s simplified by cryolite essentially looks something like this:
Al₂O₃ (dissolved in molten cryolite) + Electricity → 2Al (molten) + 1.5 O₂ (reacts with carbon to form CO₂)

See? The alumina gets broken down, and out pops pure aluminum. Simple, right? Well, the engineering and chemistry behind it are incredibly complex, but the role of cryolite is fundamentally about making that difficult separation achievable.
A Touch of History: From Greenland to Global Domination
The discovery and utilization of cryolite were absolutely pivotal in making aluminum a truly accessible material. Before the Hall-Héroult process, aluminum was actually rarer and more expensive than gold! It was considered a precious metal, used for jewelry and ornate items by the ultra-rich. Imagine a world where your aluminum foil was a luxury item!
Napoleon III of France, for instance, famously had a banquet set with aluminum cutlery for his most honored guests, while others had to make do with gold. That’s how exclusive aluminum was!
The advent of the Hall-Héroult process, powered by cryolite, changed everything. Suddenly, aluminum could be produced in vast quantities at a much lower cost. This paved the way for its widespread use in industries like transportation, construction, and packaging. From a rare gem to an everyday essential, aluminum’s journey is a testament to human ingenuity, and cryolite is a silent, yet crucial, hero in that story.
Cultural Connection: Think of the iconic Eiffel Tower. While its primary material is wrought iron, imagine if aluminum had been readily available in the late 19th century. Its lightweight yet strong properties might have made it an equally attractive, if not more so, option for such ambitious engineering feats. The history of materials is deeply intertwined with the history of innovation.
.svg/800px-Cryolite_extraction_of_aluminium_(labeled-en).svg.png)
The Modern Landscape: Synthetic Cryolite
Now, here’s an interesting twist. While natural cryolite from Greenland was historically the primary source, the demand for aluminum grew so immense that natural deposits alone couldn't keep up. This led to the development of synthetic cryolite. This manufactured version is produced chemically and serves the same essential purpose in the Hall-Héroult process.
The chemical formula is the same, Na₃AlF₆, but it's created in large industrial facilities rather than being mined. This ensures a consistent and abundant supply, allowing the global aluminum industry to thrive. It’s a great example of how science and industry adapt to meet the demands of a modern world.
Why Not Just Use Something Else?
This is the million-dollar question, isn’t it? If cryolite is so important, have scientists looked for alternatives? Absolutely! The search for more efficient and environmentally friendly methods is ongoing. However, cryolite (both natural and synthetic) has proven to be remarkably effective and economical for the Hall-Héroult process. Its specific combination of properties – its ability to dissolve alumina, its conductivity when molten, and its stability – makes it a tough act to follow.
Other fluoride salts have been explored, but none have quite managed to match cryolite’s performance and cost-effectiveness across the board. It's a bit like finding a perfectly balanced recipe; you can substitute ingredients, but sometimes the original is just… right.
Practical Tip: When you’re recycling aluminum cans (which is super important!), remember that you’re contributing to reducing the need for primary aluminum production. This, in turn, lessens the demand for the energy-intensive processes that rely on materials like cryolite. So, give yourself a pat on the back next time you toss that can in the recycling bin!
The Environmental Footprint and Future
It’s important to acknowledge that while cryolite is essential, the aluminum extraction process itself is energy-intensive and has environmental considerations. The Hall-Héroult process requires a lot of electricity, and the carbon anodes are consumed. This is why aluminum smelters are often located near hydroelectric power sources, aiming for cleaner energy.
Researchers are continuously looking for ways to improve the process, reduce energy consumption, and minimize emissions. This includes exploring new materials and technologies. However, for the foreseeable future, cryolite remains a fundamental component of how we get our everyday aluminum.
A Little Bit of Wonder
Isn't it fascinating? That incredibly light, strong, and shiny metal that’s so integral to our lives owes so much to a relatively obscure mineral. It’s a story of how a specific chemical property, found in a particular rock, unlocked a world of possibilities for humanity. It’s a reminder that even the most commonplace items often have a deeply complex and intriguing origin story.
From the ancient Egyptians who marveled at its rarity to the engineers of today who rely on its properties, aluminum has had a remarkable journey. And at a crucial juncture in that journey, a mineral named cryolite played the starring role, enabling us to build, to fly, and to connect in ways that were once unimaginable.
Reflection: The Invisible Enablers
Think about your morning routine. That coffee mug, perhaps ceramic but often with a metal lid or handle? The toaster that warms your bread? The car keys in your pocket? Aluminum is woven into the fabric of our daily lives in so many subtle ways. We rarely stop to think about the intricate processes that bring these objects into existence.
Cryolite is one of those invisible enablers. It’s a chemical workhorse, performing a vital task behind the scenes, allowing for the creation of a material that has revolutionized our world. It’s a beautiful illustration of how specific scientific discoveries and the exploitation of natural resources, when combined with human ingenuity, can lead to profound societal change.
So, the next time you reach for that aluminum can or admire a sleek aluminum appliance, take a moment to appreciate the journey. A journey that, in part, was made possible by the remarkable properties of a mineral called cryolite, a true hero in the world of everyday materials.
